Lion Biotechnologies, Inc. (LBIO) is an emerging biotechnology company focused on developing and commercializing adoptive cell therapy (ACT) using autologous tumor infiltrating lymphocytes (TILs) for the treatment of melanoma and other solid tumors.
ACT utilizes T-cells harvested from a patient to treat cancer in that patient. TILs, a kind of anti-tumor T-cells that are naturally present in a patient's tumors, are collected from individual patient tumor samples. The TILs are then activated and expanded ex vivo and then infused back into the patient to fight their tumor cells.
ACT using TILs was developed by Dr. Steven Rosenberg, Chief of Surgery at the National Cancer Institute (NCI). Lion has acquired a worldwide, non-exclusive license for various adoptive cell therapy technologies from the NCI, and entered into a Cooperative Research and Development Agreement (CRADA) with the NCI. Pursuant to the agreement, Lion intends to support the in vitro development of improved methods for the generation and selection of TILs, develop approaches for large-scale production of TILs, and conduct clinical trials using these improved methods of generating TILs for the treatment of cancers.
Currently, Lion is also in discussions with the National Institute of Health (NIH) to license additional rights to next generation T-cell technology that may have higher potency, persist over a longer period of time, require fewer cells, and have a lower manufacturing cost.
TILs therapy is presently available as a physician-sponsored investigational therapy for the treatment of metastatic melanoma in the US. Preliminary data in small patient population have demonstrated excellent efficacy and safety profile. Durable response rates have been observed in approximately 50% of metastatic melanoma patients treated with TIL therapy with complete responses of 10%. In addition to melanoma, Lion's TIL technology is potentially applicable to all solid tumors.
The TIL Technology
Adoptive cell therapy using autologous tumor infiltrating lymphocytes (TILs) was first developed in 1980s by Dr. Steven Rosenberg, Chief of Surgery at the National Cancer Institute .
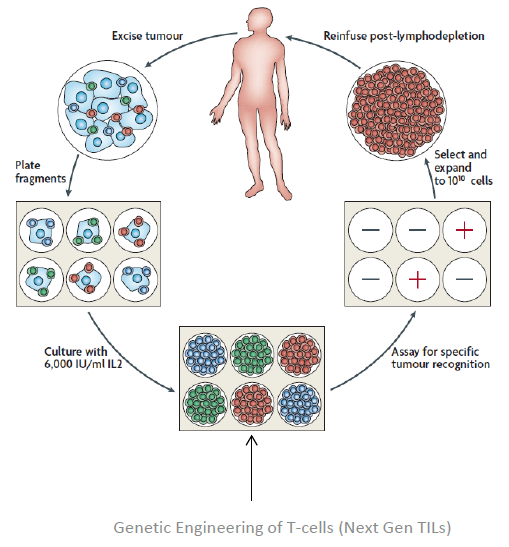
TILs are white blood cells that have left the bloodstream and migrated into a tumor. TILs, when numerous, are considered to be prognostically significant. Therapeutic TILs is a preparation of cells, consisting of autologous tumor infiltrating lymphocytes that are manipulated in vitro and, upon administration in vivo, re-infiltrate the tumor to initiate tumor cell lysis. In vitro, TILs are isolated from tumor tissue and cultured with lymphokines such as interleukin-2. The therapeutic TILs are then infused into the patient, where, after re-infiltration of the tumor, they may induce lysis of tumor cells and tumor regression. The use of therapeutic TILs is considered a form of adoptive immunotherapy.
Patients undergoing TIL therapy must have their tumors surgically resected and then shipped to the company's manufacturing facility, where the TILs are isolated, activated, and expanded to billions in vitro, away from cancer's immune-suppressing effects. These highly activated, potent TILs are then infused back into the patient, who has been preconditioned to remove all suppressive influences. The TILs are infused into the patient with interleukin-2 (IL-2) to stimulate the immune system.
TIL therapy is emerging to be a powerful treatment option for patients with metastatic melanoma. TILs together with high-dose interleukin 2 have been studied in physician sponsored multiple clinical trials in centers across the world. These physician sponsored studies have consistently demonstrated durable clinical response rates near 50% or more.
Studies also have shown that CD8+ T cells are emerging as critical for TIL therapy, although the exact subset of CD8+ T cells exhibiting the highest clinical activity in terms of memory and effector markers is still controversial.
At this point of time, a pivotal Phase II or Phase III trial is needed in an attempt to gain regulatory approval of TILs for the treatment of melanoma. Improvements in manufacturing the therapeutic TILs are also needed to reduce cost and increase efficacy. To improve response rate and duration, surrogate and predictive biomarkers are needed to better select suitable patients for TIL therapy.
We think the outlook for TIL therapy for melanoma is quite bright based on current available data. We estimate that TILs therapy will become an important treatment option for melanoma and other solid tumors in the upcoming years. Furthermore, TIL therapy in combination with conventional therapies, such as with BRAF inhibitors, as well as with Yervoy, may further increase efficacy and durable complete response rates for patients with melanoma.
Compelling Efficacy/Safety Data and Competitive Advantages of TILs
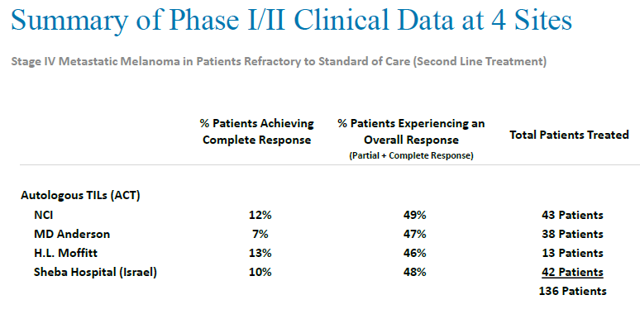
TILs therapy is a relatively new cancer therapy regimen, and is currently undergoing physician-sponsored clinical trials for the treatment of metastatic melanoma at several institutions, including the NCI, MD Anderson Cancer Center, the H. Lee Moffitt Cancer & Research Institute and Sheba Hospital in Israel.
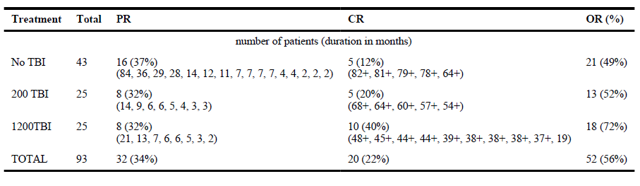
In three sequential clinical trials conducted by Steven Rosenberg at National Cancer Institute, 93 patients with measurable metastatic melanoma were treated with TILs in conjunction with interleukin-2 following a lymphodepleting preparative regimen (chemotherapy alone, or with 2Gy or 12Gy radiation). The objective response rate was 49%, 52% and 72%, respectively. Twenty of the 93 patients (22%) achieved a complete tumor regression and 19 have ongoing complete regressions beyond three years. Three and five-year survivals for the entire group were 36% and 29%, respectively but for the 20 complete responders were 100% and 93%, respectively.
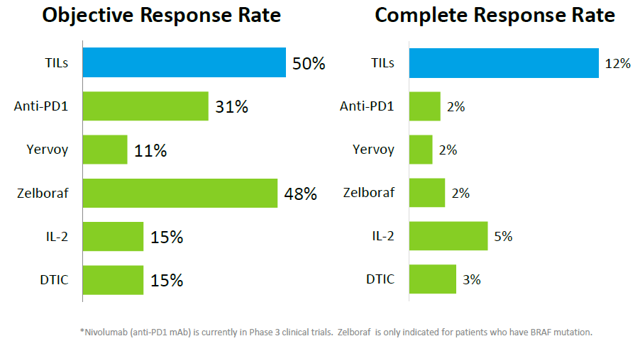
Response rate of TIL Therapy
TBI: total body irradiation
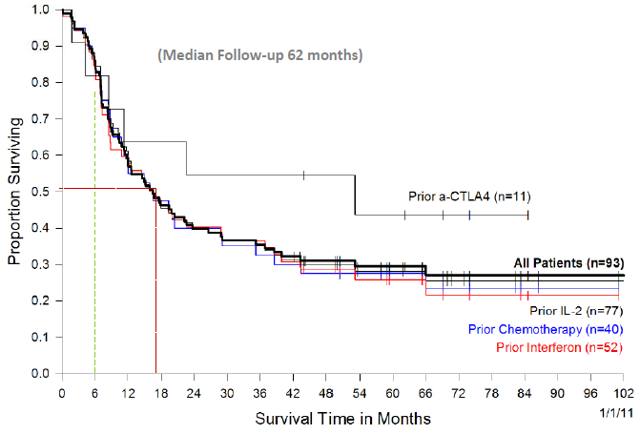
Impressive survival benefits were observed even in second line or third line setting.
Overall survival of patients receiving TIL based on prior treatment received
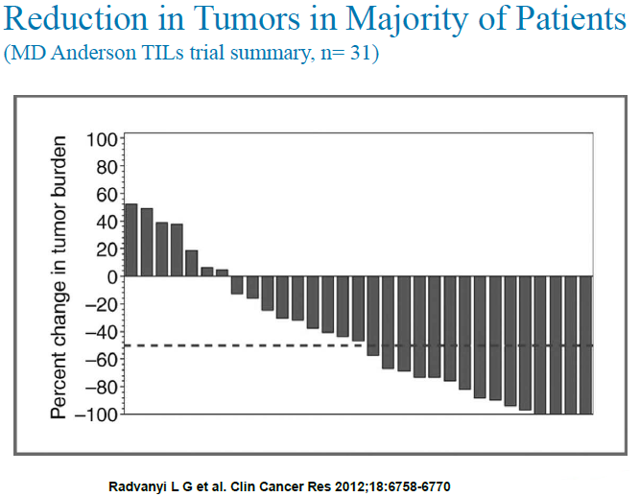
In another PI sponsored clinical trial conducted at MD Anderson Cancer Center, TIL therapy also demonstrated impressive efficacy in patients with metastatic melanoma. 31 transiently lymphodepleted patients were treated with their expanded TIL, followed by two cycles of high-dose interleukin (IL)-2 therapy. 15 of 31 (48%) patients had an objective clinical response using immune-related response criteria (irRC) with 2 patients (6.5%) having a complete response.
Of the 15 patients who responded, 12 had 70% or more reduction in their tumor burden and 4 patients had a 100% reduction in measurable tumor burden. Two patients (6.5%) had nonmeasurable bony lesions (complete response) that remained stable throughout the study period.
Progression-free survival of more than 12 months was observed for 9 of 15 (60%) of the responding patients. Factors significantly associated with the objective tumor regression included a higher number of TIL infused, a higher proportion of CD8+ T cells in the infusion product, a more differentiated effector phenotype of the CD8+ population, and a higher frequency of CD8+ T cells coexpressing the negative costimulation molecule "B- and T-lymphocyte attenuator" (BTLA).
Clinical trials conducted at other institutions achieved similar results. All clinical trials in small patient populations at different institutions show that durable response rates can be observed in approximately half of metastatic melanoma patients treated with TIL therapy. Complete responses can be seen in about 10% of metastatic melanoma patients treated with TILs.
Objective response rates are usually below 40% for existing treatment regimens approved or under clinical development. For example, objective response rates for Yervoy, IL-2 and DTIC are 11%, 15% and 15%, respectively. Zelboraf is an exception, which has an objective response rate of 48%. But Zelboraf is only approved for patients with BRAF mutation. Anti-PD1 has a relatively high objective response rate of 31%, but this candidate is still in clinical trial and its objective response rate is still lower than the 50% objective response rate for TIL.
Furthermore, complete responses in melanoma patients are extremely difficult to achieve using existing treatment regimens approved or under clinical development. Complete response rates are usually between 2-5% for most treatment regimens, way lower than the average 12% complete response rate for TIL. In Dr. Steven Rosenberg's clinical trial, TIL even achieved 22% complete response rate for metastatic melanoma patients.
Development Plan
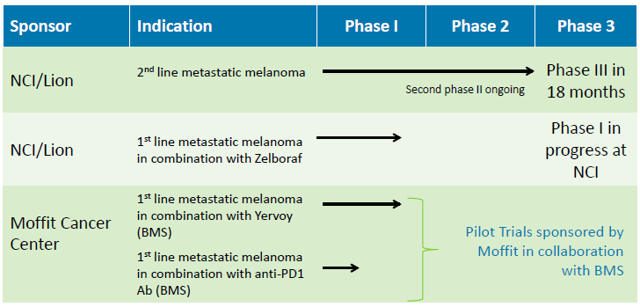
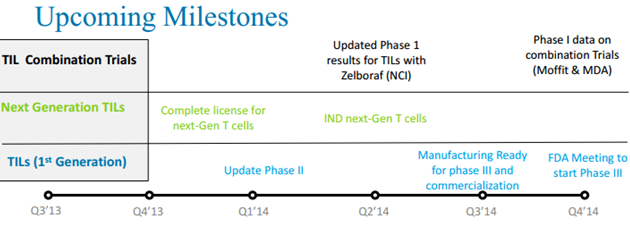
Based on the positive data from PI sponsored Phase I or II clinical trials, Lion plans to initiate a pivotal Phase III registration clinical trial to treat metastatic melanoma patients refractory to other treatments with TIL therapy. Subject to receiving FDA clearance for this trial, enrollment for the Phase III trial is expected to begin in 2015.
Lion also plans to conduct trials to treat metastatic melanoma in the frontline setting, combining TILs with CTLA-4 antibodies, such as ipilimumab (Yervoy®), BRaf inhibitors, such as vemurafenib (Zelboraf®), or PD-1/PD-L1 antibodies, such as nivolumab or lambrolizumab.
In addition to metastatic melanoma, the company intends to investigate and develop TIL therapy for the treatment of other cancers by funding exploratory pilot clinical trials under sponsored research agreements with various medical and research institutions.
TILs Clinical Trial Status
Lion is planning to optimize clinical strategies based on upcoming clinical data from PI sponsored trials. In addition to the planned clinical trials, Lion also plans to engineer a better manufacturing process for therapeutic TILs, which will generate an automated manufacturing process with minimal cellular manipulations and closed systems for manufacturing. This will increase the potency of TIL and reduce the cost of goods significantly.
Lion is also working with the National Cancer Institute to develop next generation therapeutic TILs for the treatment of cancers. There are two strategies for developing next generation TILs. The first strategy is to develop genetically engineered TILs by increasing potency, increasing persistence, and shortening manufacturing. The second strategy is to develop pre-sorted TILs by picking higher potency TILs. Both strategies lead to cheaper and better products.
Market Opportunity For TIL Therapy
The initial indication for TIL therapy will be for the treatment of metastatic melanoma. From 1975 to 2010, the incidence of melanoma tripled in the United States. The American Cancer Society estimates that about 76,690 new cases of melanoma will be diagnosed and 9,480 Americans are expected to die of melanoma in the United States in 2013.
Based on current estimates of the number of annual deaths due to metastatic melanoma, as many as 7,000 metastatic melanoma patients could be eligible for TIL therapy annually in the United States. The number of metastatic melanoma patients suitable for TIL therapy outside the U.S. is approximately twice that of the U.S. However, the number of eligible patients may significantly increase worldwide if and when TIL therapy is approved.
In addition to melanoma, Lion also plans to develop TIL therapy for other solid tumors. If TIL therapy proves to be effective to treat additional indications, the market opportunity will be significantly larger.
Patients with metastatic melanoma have a poor prognosis with a 5-year survival rate of 5%. Two commonly used therapeutic agents for metastatic melanoma are dacarbazine and interleukin-2. Dacarbazine has an objective response rate of approximately 12% with 2-3% complete response rate that are often transient. Interleukin-2 has an objective response rate of approximately 16% with 4-6% durable complete response rate.
The US FDA recently approved a few agents for the treatment of metastatic melanoma.
In March 2011, the FDA approved YERVOY (ipilimumab) from Bristol-Myers Squibb (BMY) for the treatment of unresectable or metastatic melanoma. Ipilimumab is a human cytotoxic T-lymphocyte antigen 4 (CTLA-4) antibody. CTLA-4 is expressed on activated T-cells and is a negative regulator of T-cell activation. Ipilimumab binds to CTLA-4 and blocks the interaction of CTLA-4 with its ligands, CD80/CD86. By binding CTLA4, ipilimumab enhances T-cell activation.
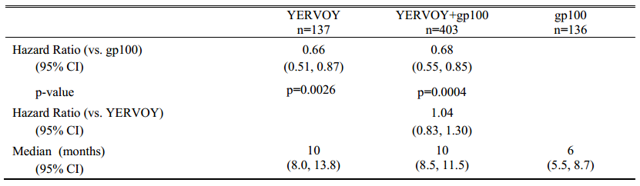
In a Phase III trial of 676 patients, overall survival was longer with ipilimumab alone compared with tumor vaccine gp100. Patients treated with ipilimumab alone had a median OS of 10 months. Patients treated with gp100 had a median overall survival of 6 months. Patients who received ipilimumab plus gp100 had a median OS of 10 months. Patients treated with ipilimumab alone also had the best overall response rate (investigator assessed), of 10.9 percent. Patients treated with the combination of ipilimumab plus vaccine arm had an overall response rate of 5.7 percent. The patients treated with vaccine gp100 alone had an overall response rate of 1.5 percent.
Overall Survival Data for Yervoy
On August 17, 2011, the FDA approved vemurafenib tablets (ZELBORAF, made by Hoffmann-La Roche Inc.) for the treatment of patients with unresectable or metastatic melanoma with the BRAFV600E mutation. ZELBORAF is a kinase inhibitor indicated for the treatment of patients with unresectable or metastatic melanoma with BRAFV600E mutation.
The confirmed, investigator-assessed best overall response rate was 48.4% for ZELBORAF. There were 2 complete responses (0.9%) and 104 partial responses (47.4%).
On May 29, 2013, the FDA approved dabrafenib (Tafinlar™ capsule) and trametinib (Mekinist tablet), from GlaxoSmithKline (GSK), for the treatment of patients with unresectable or metastatic melanoma. Dabrafenib is approved for BRAFV600E mutation, while trametinib is approved for BRAFV600E or V600K mutation.
The investigator-assessed objective response rates were 52 percent for patients treated with dabrafenib, which included a 3 percent complete response rate. The objective response rates were 22 percent for patients treated with trametinib. None of the 40 patients achieved a confirmed complete response.
The very small number of durable complete response rate makes it unlikely that many patients with metastatic melanoma will be cured utilizing any of these approaches.
Lion's TIL therapy is a totally different treatment regime for melanoma. There are several advantages to TIL therapy. Lymphocytes can be obtained from the patient, and a large number of lymphocytes can be selected in vitro for high reactivity against tumor antigens, and grow in vitro under conditions that overcome negative factors that normally exist in vivo. It's possible to modify the host prior to the cell infusion to eliminate immune regulatory cells and provide an optimal microenvironment for the infused cells.
TIL has demonstrated compelling efficacy and safety profile in human clinical trials. TIL therapy can also be used in combination with other treatment regimens for melanoma and other solid tumors. We see a blockbuster potential for TIL if approved.
Balance Sheet Boosted With Recent Financing
On November 6, 2013, Lion completed a private financing with institutional and other accredited investors for gross proceeds of $23.3 million. After paying the placement agent fees and estimated offering expenses payable by the company, Lion Biotechnologies received net proceeds of approximately $21.6 million.
At the closing, the company issued 3,145,300 shares of common stock, 17,000 shares of its new Series A Convertible Preferred Stock, and warrants to purchase a total of 11,645,300 shares of Common Stock.
The purchasers of common stock received warrants to purchase the same number of shares of common stock as such investors purchased in the Private Placement, and the investors who purchased shares of Series A Preferred received warrants to purchase the number of shares of common stock into which the Series A Preferred is initially convertible. The purchase price of each common stock/Warrant unit was $2.00, and the purchase price of each Series A Preferred/Warrants unit was $1,000. The Series A Preferred is convertible into shares of common stock at any initial conversion price of $2.00 per share.
This financing not only boosts the company's balance sheet, but also validates the clinical results to-date and the potential for tumor-infiltrating lymphocyte technology.
With the increased cash, Lion should be able to take the lead T-cell program into a Phase III clinical study for metastatic melanoma as well as sponsor several additional combination studies of check point inhibitors and T-cells.
Valuation Attractive At This Point
We are initiating coverage of Lion Biotechnology with an Outperform rating. Our 12-month price target is $20.00 per share.
Lion is an emerging biotech company focused on developing and commercializing TIL therapy for cancers with initial target of metastatic melanoma. TIL therapy is an emerging treatment regimen for cancer indications and has demonstrated compelling efficacy and safety profile for the treatment of melanoma.
TIL therapy is so far the most effective treatment for patients with metastatic melanoma. With higher response rates than Yervoy (ipilimumab) or IL-2, and longer durations of response than Zelboraf (vemurafenib), TIL therapy carries the potential to transform current outcomes in melanoma, while also defining the way cell-based immunotherapy gets incorporated into mainstream cancer treatment. TIL therapy also has the potential to be applied to other solid tumors.
Lion has a cooperation agreement with the prestigious National Cancer Institute to develop TIL therapy for melanoma and other cancers.
As the company continues advancing its current clinical programs, it is also developing next generation TILs and an optimized manufacturing process that will enable the production of highly potent, engineered cells at a significantly reduced cost. With a unique and versatile technology, promising clinical programs, strong intellectual property, high-profile collaborations and seasoned leadership, we believe Lion has the potential to create meaningful value for its stakeholders.
In terms of valuation, we think Lion's shares are undervalued at the current market price. Currently, Lion's shares are trading at about $10 per share, which values the company at $190 million in market cap based on 19 million outstanding shares. We think this is a discount to its peers considering the relatively strong fundamentals.
We understand that valuing a development stage biotech company is not easy. Lion is no exception. Most small biotech companies of development stage are valued from $100 million to $1 billion depending on how advanced the pipeline is and which indications the company is targeting. Lion is a middle stage development biotech company. TIL has finished multiple physician sponsored Phase I/II clinical trials and another PI sponsored Phase II trial is ongoing. Lion expects to have an end of Phase II meeting with the FDA in 4Q14 and start pivotal Phase III trial of TIL for metastatic melanoma in 2015. The market is huge for TIL, even for melanomas alone. If we consider other solid tumor indications for TIL, the market for TIL will be significantly bigger.
Our price target of $20 values Lion at $380 million in market cap, which we think is still conservative.
Disclosure: I have no positions in any stocks mentioned, and no plans to initiate any positions within the next 72 hours. I wrote this article myself, and it expresses my own opinions. I am not receiving compensation for it (other than from Seeking Alpha). I have no business relationship with any company whose stock is mentioned in this article.