Diabetes is responsible for $1 out of every $9 spent on healthcare in the United States. That’s 11%. 500 million people in the world have the disease. That’s 7% of the Earth’s human population.
Despite the gargantuan numbers, there is little investor excitement in the field because the big players like Novo Nordisk (NVO), Sanofi (SNY) and others dominate the market with little threat from upstarts, and the competition between these giants generally has to do with who can come up with slightly better versions and combinations of an essentially unchanged parenteral insulin treatment paradigm.
The major difference between diabetes and almost any other niche market in healthcare is that the diabetes market can be changed fundamentally just by altering the insulin administration route.
Generally speaking, making an injectable therapy orally available increases sales, patient compliance, outcomes, etc. But with diabetes, if an oral route to insulin administration makes it to market, it could change things in a much more fundamental way.
Right now the company closest to succeeding in this endeavor is Oramed Pharmaceuticals (NASDAQ:ORMP). Almost all other companies, including just recently Novo Nordisk, have given up.
Why Oral Insulin Can Change the Diabetes Market Fundamentally
Diabetes is so huge because treatment gets more and more expensive and difficult as the disease progresses, no matter how well the condition is managed. This is due to insulin injections not perfectly mimicking how insulin is stored and released in healthy people.
Think of the diabetes/insulin interplay as a game of bumper bowling. The bumpers keep the ball in the lane and out of the gutters, or blood sugar within a certain range. The tighter that range is controlled, the less damage you’ll have on the bumpers and the ball over time. But if the ball keeps smacking into the bumpers and zagging back and forth, you’re going to have a very damaged lane after a while.
The reason an oral formulation of insulin can change everything is that parenteral (injectable in this case) insulin does not communicate with the liver, whereas oral insulin, and the insulin in healthy people, does. The liver (not the pancreas, which merely produces the insulin but does not regulate its release into the blood) is the prime organ for regulating blood sugar. It stores 80% of the insulin that initially reaches it from the pancreas for later use, and based on how much insulin the liver has, it decides how much glucose to release and how much to absorb. Insulin by injection goes into the bloodstream all at once, leading to hypoglycemia if the dose is wrong. It also does not stop the liver from producing more glucose because the liver does not know how much insulin is in the blood. It only knows how much insulin is in itself.
The lack of communication between parenteral insulin and the liver means that it is a crude way of regulating blood sugar. It’s the only way we know how, but it’s still crude.
Oral insulin, if it can be done, would communicate with the liver directly, since the small intestine absorbs nutrients through the portal vein to the liver. This wouldn’t simply be a matter of convenience of administration. It would put insulin and the liver on the same page, hopefully achieving tighter blood sugar control than is possible today.
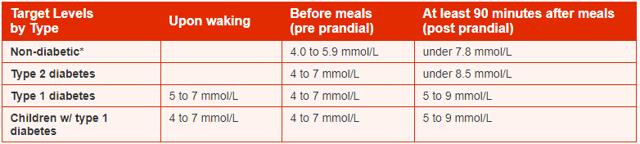
This table, copied below and taken from the National Institute of Clinical Excellence [NICE] compares recommended blood sugar ranges for diabetics versus what they generally are for healthy people. Even under perfect diabetes management, NICE knows that diabetics cannot hope to achieve the tight blood sugar ranges of healthy people.
How Good Does Oral Insulin Have to Be?
Oramed is currently on track to start a pivotal Phase III trial by end of this year or early next. An End of Phase II meeting is scheduled for August 31 with the FDA, after which a date for the beginning of the Phase III trial should be set.
The primary endpoint will probably be change in HbA1c, the gold standard endpoint in diabetes trials which measures blood sugar average over 2-3 months. The previous Phase IIb trial that Oramed conducted found that its oral insulin formulation ORMD-0801 was able to lower HbA1c in a statistically significant way after 29 days. The Phase III will be able to take a much better measurement over a longer period of time, crucial because HbA1c measures over about 90 days and not 29. The difference was slight but there, with HbA1c lower by 5% compared to placebo. Continue that for two more months and the difference may rise to 15%.
There’s also a mitigating factor here. ORMD-0801 is not Oramed’s attempt to replace parenteral insulin. The company understands that this is a nearly impossible goal. Since much of the insulin is destroyed in the GI tract, oral probably can't replace injections. Rather, the goal is only to improve blood sugar control.
That said, the setup of the Phase III trial is unlikely to be injectable insulin versus ORMD-0801, since the injectable will almost certainly win compared to oral alone. Rather, it will probably be ORMD-0801 vs placebo in patients not yet taking any parenteral insulin, as the Phase IIb was.
Phase II Results
What we know is that in patients not yet taking insulin, ORMD-0801 improves blood sugar control over placebo. Below are Phase II results taken from its investor presentation.
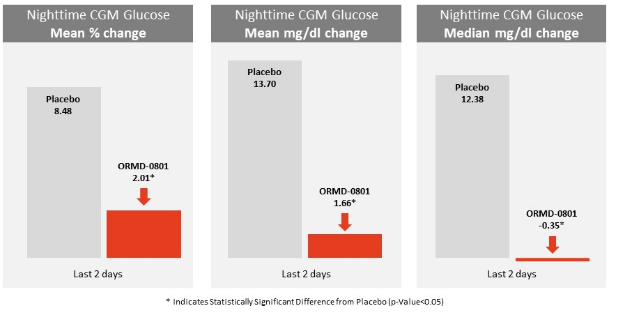
As can be seen above, the mean percent change in nighttime glucose levels was 8.48 on placebo, and only 2.01 for ORMD-0801, meaning the range in blood sugar levels was much smaller in ORMD-0801. The mean change in mg/dl was 13.7 on placebo, and a much smaller 1.66 for ORMD-0801. The median change was even smaller and actually went down at -0.35 mg/dl for ORMD-0801 and rose 12.38 for placebo.
So while the mean percent change and mean mg/dl change did rise for ORMD-0801 patients, it rose much less than placebo. That means the oral insulin is working, and can help lower the blood sugar range of pre-insulin diabetics at night. This could help delay the need for injectable insulin, and fundamentally alter the costs associated with long term diabetes care.
What about blood sugar control during the day? Results were even more impressive.
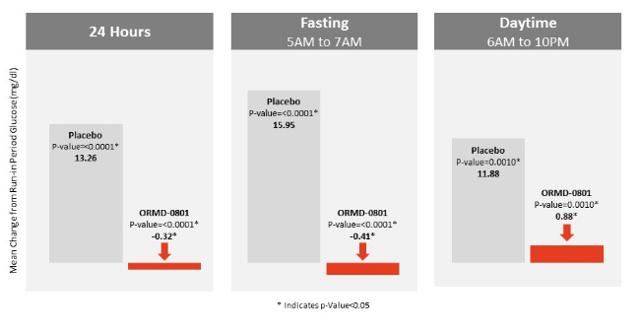 Naysayers will point out that ORMD-0801 did not lower blood sugar during the daytime (see column 3). That is true, but that is not the point. The point is that it greatly lowered the change in blood sugar during the day time for patients not yet taking insulin at all. This means it could delay the need for injectable insulin.
Naysayers will point out that ORMD-0801 did not lower blood sugar during the daytime (see column 3). That is true, but that is not the point. The point is that it greatly lowered the change in blood sugar during the day time for patients not yet taking insulin at all. This means it could delay the need for injectable insulin.
Why Now –Tel Aviv Stock Exchange
If the meeting with the FDA is yet to come and the Phase III hasn’t started yet, why invest now? Because Oramed began trading on the Tel Aviv Stock Exchange [TASE] on July 12 th, and the 30-day deadline for index-tracking ETFs to buy Oramed shares in order to keep current with the TASE is up this week. According to CEO Nadav Kidron, there will be several million dollars of buying by TASE-tracking ETFs. That is significant for a company with a market cap just above $100M, and could be a good catalyst to get in sooner rather than later.
Cash and Risk
Cash is not a pressing issue for Oramed, at least not yet. The latest balance sheet shows $1.7M in cash, $21.4M in short term deposits, $3.1M in short term marketable securities, $12M in long term investments and $1.4M in long term marketable securities. This puts the total cash and equivalents at Oramed’s disposal at about $40M. An average quarterly burn of $3.14M at about 3 years, of just over 12 quarters. However, the Phase III trial is likely to increase burn rate significantly. Let’s assume burn rate doubles, Oramed still has enough cash for about a year and a half. That should bring us to the later stages of the Phase III trial, if not its completion.
Risk with Oramed though is significant. Failure of the upcoming Phase 3 trial I assess would take off about 60-70% of its market cap. It won’t be a total loss because there are other assets including an oral GLP-1 analogue, and that molecule is more stable than insulin so it could still work even if ORMD-0801 fails. The trial could fail outright for failure to lower HbA1c, but safety is also an issue.
Issues won’t arise from the insulin itself, but the technology revolves around increasing intestinal permeability so the insulin can get through. We don’t know what happens when intestinal permeability is increased over long periods of time and whether this could cause digestive problems. So even if ORMD-0801 controls blood sugar well, long term side effects may become an issue. The other issue is that HbA1c is an ambitious endpoint and may not be met even if ORMD-0801 shows efficacy with secondary endpoints like nighttime glucose change and the like. The trial could be interpreted as a failure even if the oral insulin does have efficacy.
Conclusion
This is the biggest healthcare market in the world. ORMD-0801 won’t beat currently available insulins, but it may help delay the need for them by controlling blood sugar better in patients net yet taking insulin. If it can, it will take part of that $700B US market for itself. How much remains to be seen, and if it can show a statistically significant effect on HbA1c in the upcoming Phase III, if could even be used in conjunction with parenteral insulin for better glycemic control, which would take even more of this largest of healthcare markets.