
Quick Take
Crinetics Pharmaceuticals (NASDAQ:CRNX) intends to raise $80 million from the sale of its common stock in a U.S. IPO, according to an amended registration statement.
The company is developing a pipeline of treatment candidates for rare endocrine disorders.
CRNX has achieved promising Phase 1 trial results (in healthy patients) and the firm is highly focused on its development activities.
The IPO valuation is within a reasonable range for early to mid-stage biopharmas, although the potential market sizes for its pipeline may be somewhat attenuated.
Company & Technology
The San Diego, California-based firm was founded in 2008 to research, develop and manufacture pharmaceuticals that affect peptide hormone receptors to treat rare endocrine diseases and cancers.
Management is headed by Founder and CEO R. Scott Struthers, who was previously Member Board of Directors and President at San Diego Entrepreneurs Exchange, Sr. Director and Head of Endocrinology and Metabolism at Neurocrine Biosciences.
Crinetics Pharmaceuticals has developed pharmaceuticals for the treatment of rare endocrine diseases and endocrine-related cancers. Their drugs work by targeting peptide hormone receptors, also known as endocrine G-protein coupled receptors [GPCRs].
The company is developing pharmaceutical treatments for Acromegaly, Hyperinsulinemia, Neuroendocrine Tumors [NETs] and Cushing’s Disease.
Below is the current status of the company’s drug development pipeline:

(Source: Crinetics Pharmaceuticals)
The company has two lead products, one for the treatment of Acromegaly, and one for Hyperinsulinemia. Acromegaly is a disease caused by excessive amounts of Growth Hormone [GH] after the growth plates have closed, while hyperinsulinemia is a genetic condition that produces too much insulin resulting in low blood sugar.
The company’s lead product, CRN00808, establishes a new class of oral selective nonpeptide somatostatin receptor type 2, or sst2, biased agonists designed for the treatment of acromegaly and is the first agent in its class with reported clinical results.
In its Phase 1 safety trials, the firm reported that ‘RN00808 demonstrated clinical proof-of-concept by potently suppressing stimulated GH and baseline IGF-1 in these subjects.’
The company's second lead product, CRN02481, represents new class of oral selective nonpeptide somatostatin type 5 receptor, or sst5, agonists currently in preclinical development to treat congenital hyperinsulinism, or CHI.
Investors in Crinetics Pharmaceuticals included 5AM Ventures, OrbiMed Advisors, Perceptive Advisors, RA Ventures, Versant Ventures and Vivo Capital.
Markets & Competition
According to a 2017 market research report by Zion Market Research, the total market size for Acromegaly has reached $665 million in 2016 and is projected to reach $839 million by the end of 2022. This represents a CAGR of 3.95%.
The main factors driving the market are the lack of medical treatments in developing countries and increased epidemiology for the disease due to an increase in the tumor cases, as well as increasing awareness.
North America is expected to grow at the highest CAGR during this period due to ongoing research and development for drug discovery, as well as an increase in the epidemiology of the disease, increase in the pituitary tumor cases, high disposable income and favorable reimbursement policies in the region.
Europe comes in second, followed by Asia-Pacific in third. Europe’s better healthcare government policies are likely to boost the treatment market, while Asia-Pacific is raising its awareness for the disease, mainly in Japan, China and India.
The Middle East & Africa and Latin America will have the smallest growth in this market, due to high costs and lack of awareness.
Major competitive firms that are developing acromegaly treatments include:
- Pfizer (PFE)
- Chiasma (CHMA)
- Novartis (NVS)
- Ipsen (OTCPK:IPSEF)
- Wockhardt
- Troikaa
Pharma firms that sell or are developing hyperinsulinemia treatments include:
- Teva Pharmaceuticals (TEVA)
- Eli Lilly and Company (LLY)
- Zealand Pharma (ZEAL)
- Xeris (XERS)
- Eiger (EIGR)
- Rezolute (OTCQB:RZLT)
Financial Performance
CRNX’ recent financial results are typical of clinical stage biopharmas in that they feature large R&D and G&A expenses. The firm has some grant revenues which are relatively minor and are ‘lumpy’ in amount.
Below are the company’s financial results for the past two and ¼ years (Audited PCAOB for full years):
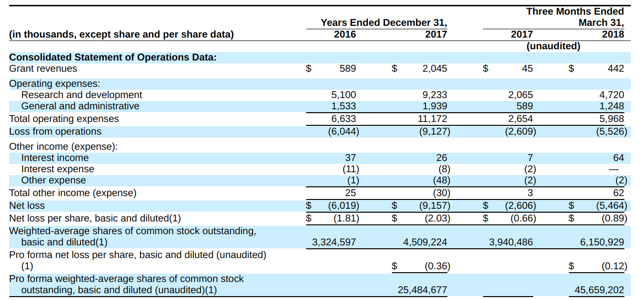
(Source: Crinetics S-1)
As of March 31, 2018, the company had $73.7 million in cash and $3.4 million in total liabilities.
IPO Details
CRNX intends to sell 5 million shares of common stock at a midpoint price of $16.00 per share for gross proceeds of approximately $80 million.
Certain existing shareholders have indicated an interest to purchase shares in the aggregate of $30.0 million at the IPO price. This is a positive signal for prospective IPO investors and is typical of life science firms going public in the current market.
Assuming a successful IPO at the midpoint of the proposed price range, the company’s post-IPO market capitalization would be approximately $352 million, excluding the effects of underwriter over-allotment options.
Management plans to use the net proceeds as follows:
approximately $45.0 million to fund the clinical development of CRN00808;
approximately $15.0 million to fund preclinical and clinical development of our other development programs; and
the remainder for working capital and general corporate purposes.
Management’s presentation of the company roadshow is available here.
Listed bookrunners of the IPO are J.P. Morgan, Leerink Partners and Piper Jaffray.
Expected IPO Pricing Date: July 17, 2018.
Editor's Note: This article covers one or more microcap stocks. Please be aware of the risks associated with these stocks.
An enhanced version of this article on my Seeking Alpha Marketplace research service IPO Edge includes:
- Valuation
- Commentary
- Opinion on the IPO
Members of IPO Edge get the latest IPO research, news, market trends and industry analysis. Start with a Free Trial.