- A 94-subject Phase 2 clinical trial, PODIUM-202, evaluating Incyte's (NASDAQ:INCY) PD-1 inhibitor retifanlimab (formerly INCMGA0012) in previously treated patients with advanced squamous cell carcinoma of the anal canal (SCAC) who have progressed after platinum-based chemo showed a treatment benefit. The results are available on demand at ESMO.
- The overall response rate (ORR) was 13.8% (n=13/94), including one complete responder and 12 partials. 33 patients showed stable cancer, implying a disease control rate (DCR) of 48.9% (n=46/94).
- Median duration of response was 9.5 months. Median progression-free survival (PGS) was 2.3 months and median overall survival (OS) was 10.1 months.
- A Phase 3 study, PODIUM-303/InterAACT 2, evaluating retifanlimab plus chemo agents carboplatin and paclitaxel in patients with locally advanced/metastatic SCAC, is in process.
- The company in-licensed global rights to retifanlimab from MacroGenics (NASDAQ:MGNX) in 2017. Incyte, in turn, sublicensed rights in Greater China to Zai Lab (NASDAQ:ZLAB) in 2019.
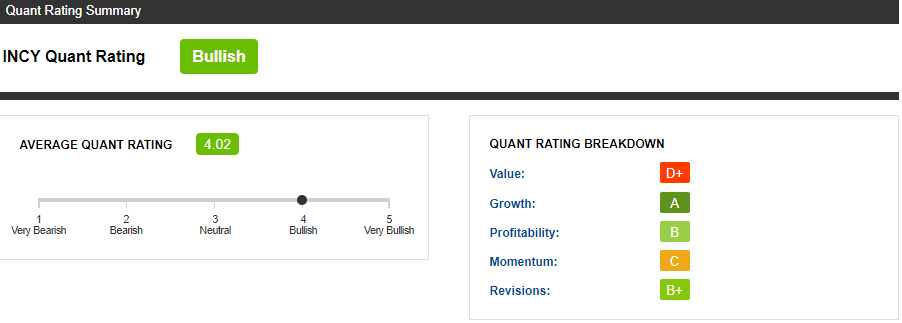
- Quant rating is Bullish.