This article discusses the corporate mission, strategies, organizational leadership, and portfolio that PharmAthene (PIP) and Theraclone Sciences will create by merging. It explains why the combination will be synergistic and increase shareholder value. Because Theraclone Sciences is a privately-held company, I give more space to its programs: I explain the importance of monoclonal antibody therapeutics and the virtues of Thereaclone's I-STAR-its engine for discovering and developing promising monoclonal antibodies . I also provide an unorthodox rationale for putting a high priority on searching for better ways of dealing with Human Cytomegalovirus (CMV) infection.
I encourage you to work through any parts of this journey that may push you beyond your comfort zone: e.g., explanations of immunological concepts, the FDA process, and the markets for the merged company's portfolio. For although Warren Buffet is right that you should only invest in companies that you understand, because I'm an educator, I believe that you are capable of grasping what the merged company will be doing, and that this article will help you do so.
Overview of the Blended Biologics Company:
Mission, Strategies, & Organizational Leadership
PharmAthene, which will retain its name and stock symbol, will become a "biologics company with extensive vaccines and therapeutics expertise" that addresses high-value markets with a focus upon infectious diseases and oncology. According to its website, PharmAthene's pre-merger mission is "to advance the Nation's health security by developing novel medical solutions that utilize cutting-edge biotechnology to offer improvements in safety, efficacy and cost-effectiveness over current therapies." According to Theraclone's website, its highly-focused mission is to turn "the repertoire's most potent antibodies into superior therapeutics." Please note:
1. While you may not fully understand Theraclone's mission (especially because of the word "repertoire"), when you learn about what its staff does, you will find that these few words communicate accurately and succinctly.
2. Theraclone's mission can be subsumed under PharmAthene's pre-merger mission by adding the six underlined words: "to restore the health of individuals and advance the Nation's health security by developing novel medical solutions that utilize cutting-edge biotechnology to offer improvements in safety, efficacy and cost-effectiveness over current therapies."
3. When you look at what Theraclone Sciences actually does and what PharmAthene aspires to do, a more accurate mission statement would replace "the Nation's health security" with the underlined phrase in this next version: "to restore the health of individuals and advance the health security of nations by developing novel medical solutions that utilize cutting-edge biotechnology to offer improvements in safety, efficacy and cost-effectiveness over current therapies."
The two companies' pre-merger strategies, strategic priorities, or activity foci can be divided into categories: operational strategies and financing strategies. While they are interrelated, it helps to focus on them one at a time.
Pre-merger operational strategies. PharmAthene's operational strategy has been to acquire and develop "new, best-in-class products and technologies identified by the United States and other governments as urgent procurement priorities." Part of Theraclone's operational strategy has been to move systematically from one development phase to the next on its lead programs. But because some of Theraclone's financing has come from working partnerships, Theraclone also focuses on executing and delivering on funded antibody discovery collaborations (as explained later).
Pre-merger financing strategies. Pharmathene and Theraclone financial strategy has been to vigorously pursue non-dilutive funding. To date, Pharmathene's funding has come primarily or exclusively from the U.S. Government. Theraclone has pursued non-dilutive funding, but has done so from two types of sources: grants (both from the U.S. Government and from private foundations) and from the abovementioned discovery collaborations. But it has also pursued funding from venture capitalists and from strategic product partnerships. When it obtains capital from venture capitalists, its strategy is make sufficient progress on its programs to enable investors to exit appropriately, such as a trade-sale (selling the company to another company) or an IPO. This merger constitutes just such an exit because its investors will collectively receive tradable shares in the merged company. When Theraclone obtains capital from working partnerships, part of its strategy is to retain key territories and/or substantial rights.
Presumably, the merged company will pursue both of the companies pre-merger operational strategies, and the following financing strategies:
- vigorously pursuing non-dilutive grants and contracts from both governmental and private sources;
- obtaining capital from working partnerships (up-front and milestone payments in the near-term, and eventually royalties);
- licensing or selling its commercial products when approved; and
- recovering expectation damages and some of its previously incurred legal costs from its litigation with SIGA Technologies (SIGA). For more information about this litigation, see here, here, and here.
Because Theraclone obtained $14 million in new funding in March, because PharmAthene's cash and accounts receivable balance at the end of the most recent quarter was $21 million, and because most of the on-going operations of both companies will be funded by grants, contracts, and milestone payments, the merged company appears to have enough funding for at least 18 months, during which it should start to receive income from SIGA. Accordingly, it should not need additional financing from venture capitalists.
The merged company's leadership team consists of an executive team and a board of directors. First I discuss the size and current affiliation of each. Next, I discuss the functions to be performed by the combined executive team members. Last, I provide background information on the new CEO and chairman of the board.
Size and current affiliation of leadership team members. The table below displays the size of the executive teams and boards of directors for the pre-merged PharmAthene, for Theraclone, and for the post-merger PharmAthene. The new executive team contains PharmAthene's three Senior Vice Presidents and all four for Theraclone's executive team members. Furthermore, PharmAthene's current CEO (see below) will retain his seat on the Board. Having a seven-member executive team for the newly enlarge company seems appropriate, given the particular functions that each will perform (see the next section).
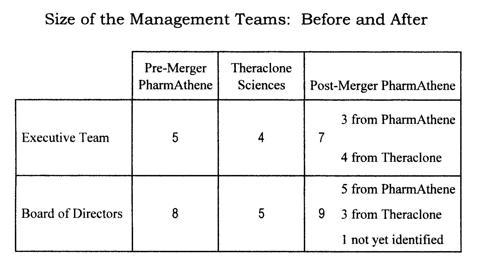 Executive functions. Three of the seven job titles held by the new executive team are quite conventional: CEO, CFO, and general counsel. However, the other four are not always found in small cap companies: chief scientific officer, chief medical officer, policy and government affairs, and regulatory affairs and quality. Three of the individuals filling these positions hold doctoral degrees, and collectively have:
Executive functions. Three of the seven job titles held by the new executive team are quite conventional: CEO, CFO, and general counsel. However, the other four are not always found in small cap companies: chief scientific officer, chief medical officer, policy and government affairs, and regulatory affairs and quality. Three of the individuals filling these positions hold doctoral degrees, and collectively have:
- expertise in microbiology, immunology, virology, and nephrology;
- an impressive track record discovering and developing protein therapeutics; and
- experience overseeing the delivery of a vaccine to the National Strategic Stockpile.
Key leadership personnel. Here are thumbnail descriptions of the merged company's top two leaders.
- The merged company's CEO. Clifford Stocks is Theraclone's current CEO. He has an MBA from the University of Chicago and has conducted academic research at university departments, one focusing on immunology, and the other on molecular genetics and cell biology. According to Theraclone's website, he recently served as Chief Business Officer of Calistoga Pharmaceuticals, where he played a leadership role in the acquisition of Calistoga by Gilead (GILD). Before that, he served as VP of business development at ICOS Corporation, where he led acquisitions and joint venture activities, as well as alliance formation, strategy, licensing and deal making. He played a key role on the leadership team that developed and launched Cialis®, and was a key architect of the joint venture partnership between ICOS and Lilly (LLY) that led to ICOS's 2007 $2.3 billion acquisition.
- The merged company's chairman of the board. Mitchel Sayare is PharmAthene's current chair. He has a Ph.D. in biochemistry from a medical school. According to PharmAthene's website, Dr. Sayare recently served as Chair and CEO of ImmunoGen (IMGN), which conducts R&D on antibody-based cancer therapeutics. While there, he led a successful IPO, raising over $300 million in venture capital and public equity financing. Before that, he served as a VP for development at Xenogen, Inc., a biotechnology company specializing in monoclonal antibody-based diagnostic systems for cancer. A later section explains what monoclonal antibodies are, and why they are important. At this point, it is enough to be aware that both the CEO and the chairman of the board of the merged company will have substantial experience with monoclonal antibodies.
And here are thumbnail descriptions, taken from the same two sources, of two key players in the current organizations, who will both be part of the merged board.
- PharmAthene's current CEO. Eric Richman, PharmAthene's current CEO, has an MBA and extensive experience in developing and commercializing novel biotechnology-based therapeutics. He was part of the founding team at MedImmune, holding various positions involving finance, strategic planning, and marketing. He was responsible for the U.S. launch of its first commercial product and was on teams that launched other products domestically and internationally. MedImmune has since been acquired by AstraZeneca (AZN).
- Theraclone's founding board member and current chair. Steven Gillis has a Ph.D. in immunology, and focuses on evaluating new life science technologies. He was a founder and director of Corixa Corp., which has since been acquired by GlaxoSmithKline (GSK). Prior to starting Corixa, Dr. Gillis was a founder and director of Immunex Corp, which has since been acquired by Amgen (AMGN). Both Corixa and Immunex focused on biologics and vaccines.
Collectively, these four individuals have impressive track records of providing shareholders with value, substantial expertise in immunology in general, and deep corporate experience with R&D involving monoclonal antibodies.
The Merged Company's Portfolio
This section will be divided into four parts: The science and business of monoclonal antibodies, Theraclone's discovery engine, the FDA approval process, and product pipelines and market
The Science and Business of Monoclonal Antibodies
How do our immune systems fight germs? With proteins called antibodies: They play a key role in sorting encountered objects-such as bacteria, viruses, and fungi-into "friend" or "foe" so that our immune systems can mount an attack against those classified as "foe." When encountering a germ, our immune system inspects a part of the germ called an antigen in order to determine whether its memory bank of antibodies contains any that has a part that fits together with part of the antigen. It is common to speak of the part of the germ's antigen as a key and the part of the antibody that fits with it as a lock, but it would make more sense to me to call the part of the germ's antigen a lock and the part of the matching antibody a key, but I did not make up this analogy. To make the analogy work, picture the lock as surrounding the key to prevent it from behaving indiscriminately. In other words, the lock acts just like a chastity belt for men.
If the immune system locates an antibody whose lock fits an antigen's key, the antibody can bind itself to the antigen, thereby either neutralizing the germ (e.g., by blocking a part that is essential for the germ's invasion or survival), or tagging it for attack by other parts of the immune system. If our immune system can not locate such an antibody in its memory bank, it creates one, and the new one joins the memory bank. In both cases, if the immune system is sufficiently robust and not otherwise distracted by other on-going threats, it works with its memory cells to generate additional copies of the matching antibody to neutralize enough of the invading the germ cells to restore health. A more thorough introduction into how the immune system works can be found here.
Unfortunately, germs are not the only villains. Tumors threaten our health, but our immune system does not declare them "foes." It fails to do so because (1) its procedure for distinguishing between friend and foe amounts to determining whether they are "self" of "non-self," and (2) tumors are our own cells. In other words, because tumors are viewed as self, they thereby avoid attack. However, if antibodies can be created or discovered that can bind the tumors, they can be introduced into our bodies where they can teach our memory cells how to generate their own copies.
The promise of preventing and treating diseases has led Pharmaceutical companies to develop monoclonal antibodies (mAbs), which are sets of duplicate antibodies made by identical immune cells that are clones of a unique parent cell. Each mAb in a set will bind to problematic cells that have the same key. It is believed to be possible to produce mAbs that specifically bind to almost any substance, including tumors. Accordingly, pharmaceutical companies are creating or searching for mAbs to attack not only infectious agents, but also cancerous cells.
There are six important criteria for assessing the utility of a given mAb:
1. Safety. Does the mAb produce any adverse effects, in either the short term or the long term? If so, with what proportions of patients, and with what severity?
2. Durability. Does it remain in our bodies long enough to continue to combat the targeted germ, which sometimes beats an initial retreat, only to rebound later when it hopes the coast is clear. It can take up to 14 days for "memory cells" to generate additional copies. Then, following another encounter with the same antigen, there will be a secondary response, which is more rapid because it activates these previously generated memory cells.
3. Immunogenicity. Does it provoke the immune system to produce antibodies against the mAb. In other words, does it ultimately generate "anti-antibodies" that might undermine its effectiveness or create other havoc?
4. Specificity. How good is the match between the mAb and the target antigen?
5. Affinity. How strong is the binding between the mAb and its target antigen? Clearly, an antibody's affinity depends upon its specificity.
6. Effectiveness. If it passes the other tests, does it do what it is supposed to do: protect against, eliminate, or reduce infections, inflammation, or tumors.
To date, Pharmaceutical companies have developed four different types of mAbs. The most recently developed type are fully human mAbs. Compared to the other three types, fully human mAbs tend to be safer, last longer, and less likely to trigger the production of anti-antibodies. So far, the FDA has approved at least 36 mAbs, but there are now about 30 available, since some have been removed because, although they were approved, their profiles on the first three criteria were not optimal. Ten the 30 that are still available are fully human mAbs, and none of those removed were fully human. The FDA approved one fully human mAb in 2002, one in 2006, four in 2009, and four since then.
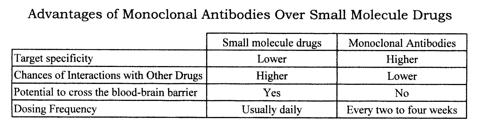
For further details, see Foltz et al., which is also the source of the information for the table below, which displays some of the advantages of mAbs over small molecule drugs.
Monoclonal antibodies are the fastest growing source of new prescription medicines. In 2012, according to William Boyle, the seven top-selling mAbs (only two of which were fully human) collectively grossed $50 billion, and the figure for the rest of the world was even larger. Of the top 20 selling biotechnology drugs, eight are mAbs.
When large pharmaceutical companies make deals with small companies that focus on discovering mAbs, the average and median up-front payments are $7.2 million and $3.6 million, respectively. The real money comes from meeting various milestones and from royalties, which average 8 to 12 percent (For further details see here).
I-STAR: Theraclone's Discovery Engine
So far, pharmaceutical companies have developed technologies to create mAbs for particular targets, to refine and optimize them, to mass produce particular mAbs that have been created or found, and to discover already existing antibodies that have highly desirable biological properties. Theraclone Sciences has pioneered a unique approach to the last of these. It recruits vaccinated individuals and immune individuals whose immune systems have, despite their history of exposure to particular germs, effectively either protected them from being harmed by them or helped them survive and defeat the germs.
It then rapidly screens tens of thousands of memory cells in these healthy individuals' antibody repertoires to find and analyze those with the highest potential for protecting against or combatting particular threats. Whereas most other technology platforms create or search for antibodies that are likely to bind with a particular antigen target and then use additional technologies to optimize their prototype, Theraclone uses a natural selections strategy that isolates those that already have high specificity and affinity. It uses the results of its screening and analysis to generate a recombinant cell line.
Theraclone's website provides this plain language summary: "Of the millions of antibodies produced by the human immune system, Theraclone Sciences' comprehensive screening technology rapidly identifies the rare few that have evolved to successfully ward off disease. We are working to develop these human monoclonal antibodies as safe and effective therapeutic products." Now, Theraclone's succinct mission statement may seem more comprehensible: to turn "the repertoire's most potent antibodies into superior therapeutics."
So far, Theraclone has focused its efforts with I-STAR in the areas of infectious disease, inflammation, and cancer. It has done so in two ways: to partner with larger pharmaceutical companies to screen candidate mAbs they will develop and commercialize, and to identify candidate mAbs that it will take the lead in commercializing (see details in the section on product pipelines and markets). Its merger with PharmAthene should allow the combined company to invest more heavily in in-house development and commercialization.
The FDA Approval Process
The FDA general approval process had a pre-clinical phase, three clinical phases, and a post-approval phase. The preclinical phase (sometimes called Phase 0) is done in the laboratory and/or in animals. For mAbs, it focuses on pharmacodynamics (what the mAb does to the body) and pharmacokinetics (what the body does to the mAb). Essentially, the preclinical phases assess durability, immunogenicity, specificity, and affinity.
The remaining phases all focus on humans, with certain exceptions described below. Phase I focuses on safety, and usually involves a small group of people (20 to 80). It determines a safe dosage range and identifies side effects.
Phases II and III focus on efficacy. Phase II, usually involving 100 to 300 people) often pits the vaccine or treatment against a placebo. Phase II is sometimes is divided into a, Phase IIa, which assess dosing requirements, and a Phase IIb, which assesses the effectiveness of the product at the prescribed dose(s). Phase II is also a more robust evaluation of the product's safety.
In Phase III trials, the product is given to large groups of people (1,000 to 3,000) to confirm its effectiveness, once again monitor its side effects, compare it to other products used for the same purpose, and collect data to guide safe use. If Phase III is successful, the product will receive FDA approval, but its use continues to be monitored during a final phase to gather additional information regarding the vaccine or treatment's risks, benefits, and optimal uses.
The standard FDA approval process described above is not suitable for certain situations. One is when individuals are becoming infected rapidly, the infections seem highly lethal, and where there are studies that demonstrate that a particular vaccine or treatment is reasonably safe in humans and appears likely to be effective but has not gone through human efficacy trials (Phases II and III). In this situation, some argue that the consequences of not providing the promising vaccine or treatment may be more dire than those of providing it. In 1992, after ten years of experience with AIDS, the FDA issued new regulations to promote the development of treatments for AIDS based on "surrogate markers," which are biological predictors for the actual target clinical endpoints. It reasoned that if it could be demonstrated that patients surrogate marker might respond rapidly to a promising treatment, it would be unethical to wait as many years as it would take to evaluate the effect of the treatment on the long term survival of AIDS patients.
Another situation where the standard FDA is process did not seem suitable involves protecting healthy citizens or soldiers from biological, chemical, radiological, or nuclear threats. If the FDA required that all promising vaccines or treatments had to undergo human efficacy trials, then it would be necessary to commit the unethical act of exposing healthy individuals to lethal or permanently disabling substances. In 2002, after not only the 9/11 terrorist attacks but also (1) our experience during the Gulf War over 20 years ago, and (2) the fact that Iraq had used chemical weapons against Iran and developed biological weapons, the FDA developed the animal rule. In brief, under certain circumstances, it allows studies that demonstrate that a vaccine or treatment is effective in animals to substitute for human efficacy studies.
Two fascinating articles-one by former FDA employee Paul Aebersold and the other by Elie Dolgin--describe the history of the animal rule and explain some of the internal debates about the validity of the principles involved. Both discuss ABthrax (Raxibacumab), which only eight months ago became the first mAb receiving FDA approval using the animal rule, and also the first mAb receiving FDA approval for antibacterial use. In particular, it is licensed for use as an adjunct to conventional antibiotics. Because ABthrax, which was developed by Human Genome Sciences, and is now owned by GlaxoSmithKline, has been included in the National Strategic Stockpile to combat anthrax, it will be discussed further in the section focusing on PharmAthene's pipeline.
Product Pipelines and Markets
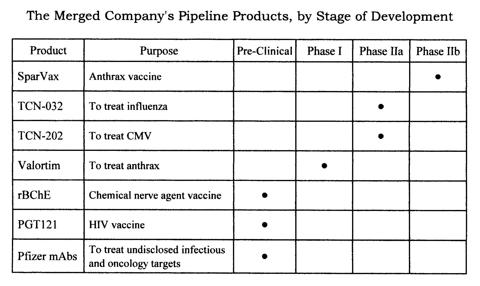
PharmAthene's pipeline contains three products, two of which are being developed to protect against and treat anthrax. The third is being developed to protect against a full array of chemical nerve agents. Before discussing the specific products, we shall take a quick detour to look at competing products as well as possible markets beyond national governments.
A perspective on competing products and possible. Since the 9/11 attack, spores have been mailed to newscasters and politicians, and five people had died from inhalation anthrax. PharmAthene correctly argues that BioThrax, the only FDA-approved vaccine, uses 60 years-old and inefficient technology. However, BioThrax's owner, Emergent BioSolutions (EBS), is constructing a modern facility that it hopes will be licensed in 2015 that has the capacity to produce up to 25 million doses per year. Furthermore, EBS has conducted recent tests on BioThrax's ability to be administered after exposure, and is also developing three other anthrax products, one of which combines BioThrax with an mAb adjuvant.
How problematic to PharmAthene is the fact that there are anti-anthrax products already approved or being developed by Emergent BioSolutions and GlaxoSmithKline? In his previously cited article, Aebersold quotes a scientist who had been working on ABthrax as pointing out that the Animal Rule's preamble:
1. "describes the need for a wide range of therapeutic options for the treatment of bioterror pathogens and specifically cites anthrax,"
2. states that the existence of a proven treatment for a given toxic agent does not constitute a barrier for approval, an
3. clarifies that it is not necessary to demonstrate that, compared it existing products, new protection products provide superior benefits.
In other words, the Federal Government is committed filling the Strategic National Stockpile with a variety of products that protect against or cure anthrax (and other biochemical terrorism threats). Less than two months ago, it affirmed this commitment by lifting the hold that it had placed on the SparVax Phase II trial (which had not yet started).
Regarding markets, PharmAthene has historically been focused on the biodefense market: BARDA and foreign governments. However, since humans can catch anthrax from animals, there may be other American customers, both within and beyond the government. Since Theraclone Sciences has already been planning to sell products to this full range of customers, PharmAthene will now be re-positioning itself to do the same.
Without further ado, here are PharmAthene's three products.
SparVax. SparVax is a highly purified recombinant protective antigen that is being developed for both pre- and post-exposure. It can rapidly be scaled-up, has enhanced convenience (because it is packaged in prefilled syringes), and has already successfully completed Phase I and Phase II trials, which demonstrated that SparVax is at least as safe and effective as BioThrax. In fact, the proportion of subjects experiencing pain at the injection sire was more than double for those receiving BioThrax as receiving SparVax.
However, because previous Phase II studies have been carried out with SparVax that was manufactured in Great Britain, the FDA is requiring PharmAthene to conduct another Phase II trial on SparVax that has been manufactured in the U.S. There is no reason to expect a different result in the U.S. trials. So far, PharmAthene has received funding of $213 million for SparVax if all milestones are met and the U.S. Government exercise all options under the contract.
Finally, like BioSolutions, PharmAthene is working to improve its product. Thus, while the testing of SparVax is continuing, PharmAthene is simultaneously experimenting with ways of combining the recombinant protective antigen that is used in SparVax with a different adjuvant in order to increase both its stability and potency.
Valortim. Although SparVax and the competing vaccines can be administered after exposure, they tend not work very well with patients who have already developed advanced infections. There are existing antibiotics and the treatment of choice is Cipro, but because it needs to be taken twice a day for 60 days, there are often patient compliance problems.
Valortim is a fully human mAb whose mechanism of action is similar to our immune system's natural response. Studies have demonstrated that a single dose will provide substantial protection in monkeys, whether administered before or after exposure. PharmAthene is developing Valortim in partnership with Bristol-Myers Squibb (BMY).
Anti-nerve agents. PharmAthene developed Protexia, which in tests on guinea pigs, demonstrated efficacy against all the chemical nerve agents tested, including VX, Sarin, Soman, and Taubin. Although animals treated by the accepted standard of care had severe neuropathy, those treated with Protexia showed no neurological damage. Protexia was shown to be safe and well tolerated in a Phase I clinical study with humans.
However, the U.S. government considered the cost per dose to be too high, so it has funded PharmAthene's shift to a more advanced and less expensive treatment that involves a human cell culture-based manufacturing system that produces a product that is identical to that generated by our own bodies. Because the next iteration is a human product, it should have a significantly longer half-life. While supporting PharmAthene's R&D for an anti-nerve agent that is more efficacious and cost effective, the U.S. Government has also increased the number of doses that it intends to procure.
Theraclone's portfolio consists of mAbs that it is developing both with and without partners. I shall start by describing Theraclone's three partners and then turn to its own products (which may be developed with non-dilutive U.S. Government funding).
The partnership with Zenaku Kogyo. The purpose of the partnership is to use Theraclone's I-STAR technology to discover broadly protective monoclonal antibodies for the treatment of pandemic influenza and severe seasonal influenza. The next paragraph describes the nature of the partnership; details about the lead product under investigation will be described in the section describing Theraclone's pipeline candidates.
According to a press release, Zenyaku Kogyo will have an exclusive license in the Japan territory to any influenza products developed under the program, and will be eligible to receive a share of proceeds from Theraclone's licensing of rights in Asian countries outside of Japan. Theraclone is eligible to receive clinical milestone payments and royalties from potential future sales in the territory, and will retain worldwide development and commercial rights outside of Japan.
The partnership with the International AIDS Vaccine Initiative. Since I have not been able to locate information about the nature of the financial arrangements of the collaboration between Theraclone and IAVI, which is partly financed by the Bill & Melinda Gates Foundation, I shall provide some details about the partnership's purpose and results to date. These details demonstrate how I-STAR works.
Broadly neutralizing monoclonal antibodies (bnMAbs) derived from rare individuals with protective immunity against HIV-namely, HIV-infected but long-term non-progressing patients-provide clues to important HIV antigens. I-STAR has been used to examine the memory cell repertoires of such patients and has identified at least 16 highly potent targeted antibodies. IAVI is responsible for testing the vaccine candidates considered promising.
Less than a year ago, an article in the highly prestigious journal Proceedings of the National Academy of Sciences described the results from studies of PGT121, one of the bnMAbs resulting from I-STAR activity. In way of background, the standard way of discovering whether a vaccine candidate might be effective is to administer the vaccine to primates and then infect them with a large dose of HIV. Most animal tests of HIV bnMAbs have associated protection against such high-dose viral challenges with relatively high concentrations of the vaccine candidate in the blood.
The investigators administered three groups of rhesus macaques different doses of PGT121 24 hours before infecting them with simianhuman immunodeficiency virus (SHIV). Sterilizing immunity was achieved in all macaques administered the two higher doses and three of the five that received the lowest dose. The results suggest "that PGT121 can mediate sterilizing immunity at serum concentrations that are significantly lower than those observed in previous studies."
The partnership with Pfizer (PFE). According to an article in the New York Times: "Pfizer, the world's largest drug company, has agreed to pay up to $632 million to Theraclone Sciences in a research collaboration . . . [using I-STAR]. Neither company would specify the amount of initial financing for the deal. The upfront money will take the form of technology access fees on two unspecified targets in oncology and two unspecified goals in infectious disease . . . [According to Dr. Gillis], $189 million to $252 million was 'near term money' for milestones before the Phase II clinical trials. Theraclone could also earn royalties on any developed products. Pfizer is responsible for preclinical and clinical development."
So far, Theraclone has completed its obligations regarding the first undisclosed infectious disease target and is well along doing so with the second undisclosed infectious disease target. Just a year ago, Pfizer identified its first oncological target.
Theraclone's pipeline contains mAbs for two diseases: flu (both seasonal and pandemic) and CMV.
Seasonal and pandemic flu. Each year, 200,000 patients in the U.S. are hospitalized with severe cases of flu, and 30,000 die here, and between 250,000 and 500,000 die worldwide. To treat such patients, Theraclone is developing TCN-032, a mAb whose lock binds to the key in over 98% of known influenza A strains, including avian and swine flu. During a Phase I trial, TCN-032 was well tolerated and exhibited a favorable safety and pharmacokinetic profile. This fall, study team researchers will report on the completed Phase IIa viral challenge trial.
Pre-clinically, Theraclone is also screening human donors for broadly neutralizing, cross-reactive human antibodies whose lock will bind to the keys of 17 subtypes of another antigen target, known as hemagglutinin. So far, several promising candidates have been identified.
Human Cytomegalovirus. CMV is pervasive, but CMV infection is typically unnoticed in healthy people. However, it can be life-threatening for those with a compromised immune system, such as HIV-infected persons, organ transplant recipients, chemotherapy patients, and new born infants. After infection, CMV can remain latent within the body over long periods, and reactivated when the immune system is challenged. As Theraclone's chief medical officer Eleanor Ramos points out, "CMV is a difficult-to-treat indication due to the virus' propensity to mutate and become resistant to currently marketed drugs which additionally are not fully effective and are associated with significant toxicities."
Without challenging the well-established finding that CMV is one of the infections that AIDS patients develop, Robert Root-Bernstein presented compelling evidence here and here that:
- Healthy people tend not to develop HIV, and are more likely to become infected by it if the immune system is somewhat compromised.
- CMV is one of the "co-factors" that facilitates HIV infection.
- CMV is also a co-factor that facilitates the progression of HIV to AIDS.
- People with CMV have developed AIDS-like symptoms without testing positive for HIV.
To treat CMV, Theraclone is developing TNC-202, a broadly protective, fully human mAB. In a randomized, double-blind, placebo-controlled Phase 1 study involving 40 healthy adult volunteers, TCN-202 showed pharmacokinetic parameters consistent with human antibodies, and no antibodies against TCN-202 were detected. The detailed results from this trial will be presented September 10 at ICAAC 2013.
Summary
Merging PharmAthene and Theraclone Sciences creates a biologics company with an expanded set of high-value markets in the area of protection from terrorist threats, oncology, and infectious, inflammatory and autoimmune diseases. The combination of the wider focus, I-STAR, and the SIGA litigation provide both a rich variety of non-dilutive funding sources and opportunities for partnerships beyond Bristol-Myers Squibb, Zenyaku Kogyo, IAVI, and Pfizer. As a unique discover engine, I-STAR will enable PharmAthene to refill its pipeline continually.
The executive team has been creatively designed, with seven thoughtfully targeted functions. Collectively, the merged company's key personnel have impressive track records of providing shareholders with value, substantial expertise in immunology in general, and deep corporate experience with R&D involving monoclonal antibodies.
The table below shows the range, depth, and status of the combined company's portfolio.
Disclosure: I am long PIP. I wrote this article myself, and it expresses my own opinions. I am not receiving compensation for it (other than from Seeking Alpha). I have no business relationship with any company whose stock is mentioned in this article.