VistaGen (NASDAQ:VTGN) is a strange company that has been around quite a while. They've developed some stem-cell research platforms with the help of Dr. Gordon Keller, but that particular platform has never really amounted to much success. There is some hope that it could potentially provide revenues in the future, but this article will ignore it, and in total will not consider its future value.
Instead, we take a look at VTGN's lead (only) drug candidate, AV-101. The drug is similar to the endogenous kynurenine metabolite, of the kynurenine pathway.
Image created by author
Kynurenine (and 4-chloro-kynurenine, which is also AV-101), are precursor molecules to kynurenic and 7-chlorokynurenic acid, respectively. 7-chlorokynurenic acid does not have any ability to cross the blood-brain-barrier (BBB), but it is an active NMDA antagonist.
In order to get the drug into the brain, VistaGen (and previous researchers) thought to create a pro-drug, 4-chlorokynurenine, that would get into the brain, and thus be metabolized into the active form.
But we're getting ahead of ourselves. What's an NMDA antagonist and why do we care about this?
NMDA antagonists are one of the more promising, but unknown, potential drugs in psychological disorders (such as major depressive disorder, MDD). And they have really gained a lot of attention in the past decade due to the story of ketamine - the horse tranquilizer that has been around for over half a century.
Ketamine, it turns out, has a fantastic side effect of mitigating depression and suicidal thoughts. There have been multiple studies confirming this, but this is not the only side effect.
Ketamine also produces serious issues, such as loss of motor control and hallucinatory and psychotic-like symptoms, as well as problems with blood pressure. Nevertheless, the drug (or esketamine) has been making its way through clinical trials, backed by the large-cap Johnson & Johnson (JNJ) through Janssen.
Some of the stories about ketamine's ability to work through depression are rather impressive due to its fast-acting action mechanism. The lowering of depressive symptoms can outlast the half-life of the drug, which is quite remarkable, and this was a major impetus for pushing this drug into the clinic. However, in addition to ketamine's unwanted side effects, it also requires either IV or intranasal administration.
AV-101 is somewhat orally active, although the numbers aren't stellar. AV-101 itself (the prodrug) is about 100-fold higher in oral bioavailability than ketamine (based on Cmax calculations, not AUC):

Image from M. Wallace et al./Scandinavian Journal of Pain (2017)
This potentially could translate to a 100-fold increase in the actual active drug, 7-Chlorokynurenic acid, within the brain. However that isn't quite clear or supported in humans at this point.
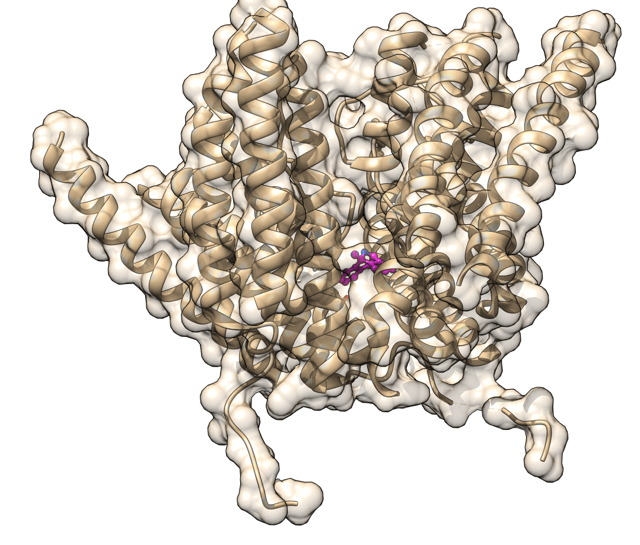
There is also a difference of binding site for 7-Cl-KYNA versus ketamine, which is why the company and researchers believe that it could potentially have all the good and none of the bad, with respect to ketamine. Pretty picture time!
Image created by author, PDB: 5UN1
This is the NMDA receptor and within it, that small pink thing is the well studied NMDA-antagonist MK-801. The antidepressant potential of MK-801 has not reached a consensus, with articles showing effectiveness exists while other says it's a dud. But that doesn't matter for our AV-101 - it doesn't bind in that site.
It binds to the 'glycine' site, which is located at the periphery - on the outside of the NMDA receptor - and thus has a different mechanism for activation and different outcome. This could potentially mitigate the extreme side effects of many of the current clinical-trialed NMDA-antagonists.
The Market Cap Question
So, unlike MRNS, which has rallied hundreds of percent by simply being a me-too to SAGE's new GABAa anti-depressant drugs, VTGN has gotten nothing short of ignored for this potential blockbuster drug. Sitting at under $30 million market cap, there have to be some other pieces to this puzzle that paint a bearish picture.
They are numerous and will require our attention.
Pain
First, the above-cited Scandinavian Journal of Pain article was about a study that attempted to show that this drug could produce analgesic like effects for pain - much the same way ketamine does - and that it has been approved for this since the 1970's.
Here's the conclusion:
Although, AV-101 did not reach statistical significance in reducing pain in the capsaicin induced-pain model, there were consistent reductions, in allodynia pain and mechanical and heat hyperalgesia.
In short, it didn't work for their pain model. But ketamine would have (or has already done so). So, a major question: if the drug can't mimic ketamine in pain analgesia, why should we expect it to mimic it in the antidepressant effects? Again, our only option is to believe that the alternative binding position is responsible (but still allows for antidepressive effects!).
Trial Design Strangeness
This is a National Institute of Mental Health-sponsored study. I have never seen anything like this, and if any reader has, please point me to it. I would be very grateful.
 Image taken from clinicaltrials.org
Image taken from clinicaltrials.org
If you can't read that, it says:
Participants will stay in the hospital for 12-14 weeks.
This study is our next catalyst, which is the only real catalyst that stands between us, dilution, and the larger Phase II study scheduled for top line results in 2019. The company hasn't been quite clear on when this readout is supposed to occur, citing everything from the end of 2017 to 1H of 2018, and most recently just 2018. As you can imagine, getting people who are depressed to sign up for a study where they will stay in a hospital for three to four months might be difficult.
However, if you're interested, travel arrangements will be made, and compensation provided. I contacted the trial coordinator, but have not heard back yet.
My issue with this is that this should be our data-driving event, but with that type of requirement, I don't see this trial ever fully enrolling. Could I be wrong? Totally. The doctor who is leading this study likely deals with chronically depressed people seeking serious help for their depression, and thus it could potentially be possible.
Finances
VistaGen doesn't have much cash. As of their most recent filing, they had about $13 million in cash, with a quarterly burn of ~$3 million. That gives us potentially 2, and if we get optimistic, 3 quarters where dilution could be avoided.
What is really outstanding is the amount of warrants at $1.50 - there are 10 million of them, which would be around 45% of the total outstanding shares currently out there.
So the risk of dilution comes from both a raise in capital through offering of common stock, and dilution fears if the stock rises above $1.50 due to warrant exercise.
Safety Issues
While the safety issues so far have not been a red flag for me in humans, some pre-clinical worries do exist. Image taken from Scandinavian Journal of Pain article
Image taken from Scandinavian Journal of Pain article
That Cmax value comes from preclinical safety concerns in dogs, reported in their 10-K:
In dogs, oral administration of AV-101 resulted in CNS-related clinical signs, including decreased activity, abnormal gait/stance, ataxia, and prostration.
The dogs lost motor control and started moving involuntarily. That dose could potentially be about double the maximal dose seen in these trials for humans. Thus - don't take two pills, please.
Final Summary
This is a strange one. At first, I was very bullish and believed I had found a hidden gem in the pile of overpriced, gilded wares of the biotech sector - a neglected company with a potential blockbuster CNS drug. The next CPXX, in my mind.
But the further I dig the more I realize why the market cap is essentially echoing a large "No" for investors:
- The science seems supported by a shaky me-too thesis (the company says the word ketamine about as often as AV-101 on their website).
- Their most objective NIMH trial is designed, seemingly, to never release data.
- Their finance overhang is heavy.
However:
There is the potential that we see some glimmer of efficacy for this ketamine-like drug. If there is any indication that this is equivalent in efficacy to ketamine, there is potential to be "the best performing biotech of 2018." Much akin to Marinus (MRNS) in 2017, the parallels are hard to ignore.
So really it comes down to timing, and timing is difficult:
- We can expect dilution - but when?
- We might maybe potentially see NIMH study results - but when?
- The company's actual self-contained Phase II isn't until 2019
With the potential for this company to catch wind, and for investors to see the parallels between this and MRNS, I have started a very small position in what I would consider my most risky investment currently in my portfolio. I would intend to slightly increase that following a dilution event.
If the data comes out at all comparable to ketamine, then I will absolutely consider a more moderate position in what would then be a true gem in the rough.
Editor's Note: This article covers one or more microcap stocks. Please be aware of the risks associated with these stocks.