Quanterix (NASDAQ:NASDAQ:QTRX) is a fledgling small-cap company with lofty aims to disrupt the biomarker market. To this end, their Simoa platform boasts the only current combination of best-in-class sensitivity, solid infrastructure, and high throughput capabilities. The company appears to be making moves to set itself up as a long-term player; however, there remain many questions as to whether drastically increased sensitivity will translate to viability in the clinic and implementation in medical applications. Additionally, the company is overvalued for its current size, and any further growth in valuation is dependent on the ability of their assays to show efficacy in indications currently lacking biomarker options.
Simoa Technology - High sensitivity digital ELISAs
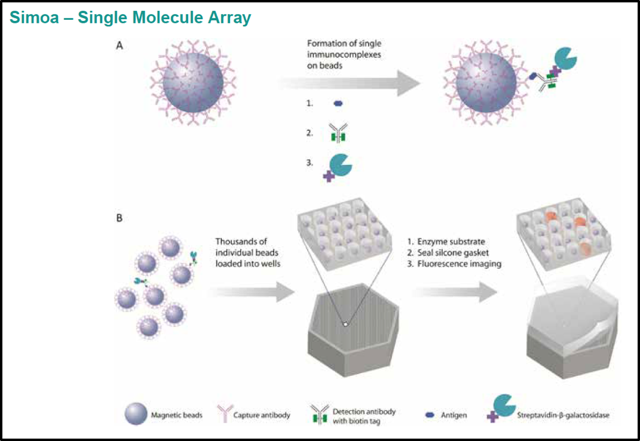
Quanterix’s flagship product Simoa, or Single Molecule Assay, is a microwell magnetic bead-based digital ELISA (Enzyme Linked Immuno-Sorbent Assay) system that is 1000 times more sensitive than current methods.
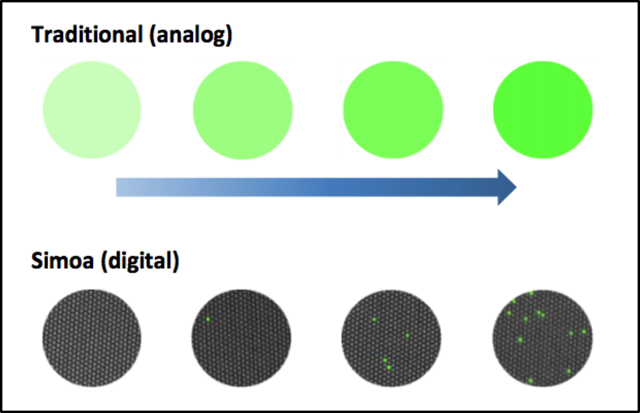
ELISAs are a near ubiquitous assay in molecular biology used to determine the concentration of analytes of interest. All ELISAS involve coupling the target analyte to biotinylated detection antibodies, which are subsequently detected by a reporter enzyme. The chemical reaction generates a fluorescent substrate which is detected and quantified. The traditional detection limits of these tools are in the picomolar concentration range due to their analog signal output.
Simoa’s innovation lies in the linking of these targets to magnetic beads which can then be distributed individually into hundreds of thousands of tiny wells sized to capture just one molecule-bead complex each. Each bead captures either a single immunocomplex or none, and subsequently are distributed into individual wells. Detection of a fluorescent signal in each well allows a binary (or digital) readout, providing a high resolution output. Through this technique, a much greater sensitivity is achieved than through traditional methods - detecting 100 to 1000 fold lower target concentrations.
The resolution achieved with Simoa can be striking - some labs have even been able to use this system to detect concentrations of analytes in single cells. A dynamic range of 4 orders of magnitude means that the technology can be used in all current biomarker diagnostic applications as well as those in the future requiring higher sensitivities.
Strategy- A focus on expanding and validating potential application spaces
Quanterix is betting that the best in class sensitivity of their Simoa technology will open new niches in the biomarker diagnostic space, in particular in neurology and oncology where there has been little success establishing medically actionable biomarkers at current sensitivity standards. CEO Kevin Hrusovsky has emphasized that Quanterix intends to drive consumer adoption through the promotion of basic research utilizing the Simoa technology at both CROs and academic institutions. To this end, Quanterix has doubled the number of publications using its product in each of the prior 3 years and is on track to double the number again this year. The hope here is that researchers will begin to uncover biomarkers with medically significant ramifications when present at the femtomolar scale. Quanterix’s customer base is currently split approximately evenly between CROs and academic institutions.
Neurofilament Light (NFL) is to date the most promising example of the type of biomarker the company is hoping their product will help identify. This protein has been found to be a decent plasma metric of axonal damage - as neurons are damaged, fragments of Nfl are released and cross the blood brain barrier into circulation. So far, this biomarker has demonstrated viability in multiple sclerosis, neurodegeneration, and most robustly, traumatic brain injury. The National Football Association has identified the protein as a possible medically actionable readout of brain injury severity and sponsored further research using Simoa technology. Multiple publications from academic researchers have corroborated these conclusions. QTRX’s assays are the only available with the capability to detect Nfl at pathologically relevant levels.
Medical adoption of any biomarker diagnostic, is of course dependent upon substantial validation of the marker in clinical trials as a reliable endpoint. Quanterix is also driving usage in this area, noting that clinical trials have a 200% increased approval rate when using a biomarker as a core quantitative metric. Nonetheless, many of the new biomarker targets being identified by Quanterix’s technology are still early along in the validation process.
Apart from new biomarker targets leveraging Simoa’s superior sensitivity, the catalogs of QTRX and its leading competitors are quite similar. Most of the assays in QTRX’s area of focus, including neurology and oncology, are also sold by their competitors, albeit with inferior sensitivity. Such sensitivities have not to date proven medically useful for these disease spaces, something QTRX believes it can overcome with its technologies. Their overall consumable line includes more than 200 target molecules and has expanded every quarter since their IPO. Additionally, earlier this year QTRX acquired Aushon, a company with a similar high sensitivity ELISA platform, providing access to Aushon’s expanded menu of assays targeting primary cytokines.
Quanterix, similar to Luminex and its other competitors, employs the razor-and-blade business model, whereby expensive machinery is sold up front at smaller margins in order to ensure a lengthy consumer relationship and increasing profits down the line as these machines are only compatible with their proprietary consumable kits. If consumers begin to transition to Simoa high-throughput machines for exclusive high-sensitivity neurology and oncology assays, they will have a financial incentive to also use Simoa consumables for all their assay needs. In this way QTRX may be able to force a space for itself in the market, even if the majority of its assays are comparable to those of competitors. However, Consumers will need a significant and medically valid reason to switch suppliers, and QTRX’s sole advantage here is in its product’s sensitivity. Quanterix’s flagship machine, the HD-1, has similar specs to Luminex’s Aries series in terms of throughput ability and ease of use.
A focus on the development of strong research foundations and a diverse consumer base, if executed successfully, may position Quanterix to maximize its share of new rapidly growing corners of the biomarker market. If these spaces fail to blossom due to Simoa’s improved sensitivity as the company hopes, however, their outlook is much dimmer. Indeed, in established biomarker indications (i.e. infectious diseases) there is little evidence that drastically enhanced sensitivity would produce clinical benefit. With a high initial cost of switching to Quanterix’s platform, there is thus a significant risk that potential customers will stick with their current providers, and QTRX may fail to capture a sufficient market share for its valuation.
Rapidly expanding market
The market for biomarker diagnostics today lies at about $32B globally. Many estimates extrapolating from current growth trends anticipate this space will balloon to $80B by 2025. Much of this market is comprised of competitors focusing on infectious disease diagnosis, and the $80B estimate does not account for emerging markets, such as oncology and neurology, which Quanterix anticipates will be valued at greater than $30B. Unless another biotech develops a parallel technology with similar sensitivities and throughput capabilities to Simoa, QTRX is positioned to corner much of the market in these new areas.
In the last three-odd years since its IPO QTRX has grown at a breakneck pace, and doesn’t appear to be slowing down. Their Q3 revenues posted- posted November 1st- showed a 64% YTD increase in revenue to $27M, with an average 37% CAGR since 2015. Product revenue totaled $6M for Q3, an increase of 82% on last year. Each of the last few quarters’ releases have painted a similar picture. As more clients are onboarded and drive increasing consumable use, the most rapidly increasing share of QTRX’s revenue, this growth can be expected to at the very least remain constant in the near future, if not accelerate further. The pace of this growth is even more rapid than that experienced by Luminex during its early years (27% CAGR), likely due to buoying from the expanding diagnostic market space. While eventually revenue increases will level out, in the short term, financial results are expected to exhibit a continuation of these recent trends.
Despite this massive market potential and strong growth, QTRX already seems to have much of its prospects baked into the current valuation. Our DCF forecasts, assuming 80% operating expenditures (Luminex’s average OpEx) and 30% revenue growth from 2019-2025, places the firm at about $4 a share given a 10% discount rate. In order to achieve the value currently set by the market (around $20 a share), the firm would need a nearly 60% growth rate over the next 10 years, a continuation of the current YTD revenue increases but at a much larger valuation size. If Quanterix’s predictions about the market’s growth and their own disruptive capabilities are believed, this is not out of the realm of possibility. However, the biotech would need many of its large bets to break perfectly to reach this goal. Until more information on QTRX’s trajectory is available, the company seems to be overvalued due to substantial enthusiasm about the exciting implications of their technology.
Strong but straggling competitive landscape
The current competitive landscape for biomarker detection presents a hurdle for Quanterix. The primary competitors- Luminex, Millipore Sigma, Biotechne, and MSD- together hold more than 80% of the market and follow similar razor-and-blade models, meaning changing suppliers for consumers requires a significant initial investment in new machinery. In the future, as Quanterix expands into a role in health screening, they can expect to compete with additional established companies such as Siemens, Abbott, Roche, Ortho Clinical Diagnostics and Thermo Fisher Scientific. These corporations are much larger and have significant momentum. Fortunately, Quanterix’s technology lends them a unique and advantageous value proposition.
Quanterix is the only player in this space that promises a combination of sensitivity, automation, and multiplexing. Independent validation has concluded that only the Erenna platform from Millipore Sigma can achieve a similar degree of sensitivity and dynamic range, yet the supplementary kits and software for this device falls short of the throughput, ease of use, and automation capabilities of Simoa’s HD-1.
While the largest competitors do not yet have the level of sensitivity required to directly compete with Simoa technology, it is feasible they may develop these capabilities in the future. However, due to the structural requirements of high-sensitivity ELISA quantitation, it is very unlikely Luminex or its peers will be able to leverage their current flagship machines for use in these applications. Momentum in this field is essential, and there appears to be a reasonable barrier to entry in high sensitivity diagnostics for the largest competing players.
Despite having a solid, growing research foundation, it remains to be seen whether the Simoa technology will prove as disruptive or medically actionable as Quanterix hopes. Much of the optimistic valuation of the company relies on the prospect of yet undiscovered applications for the company’s product. It is not unlikely the frontiers created by enhanced detection sensitivities will generate some new market arenas for QTRX’s exploitation. However, Simoa needs to prove novel efficacy in many indications for the company’s value to exceed its current lofty position.
References and Additional Information