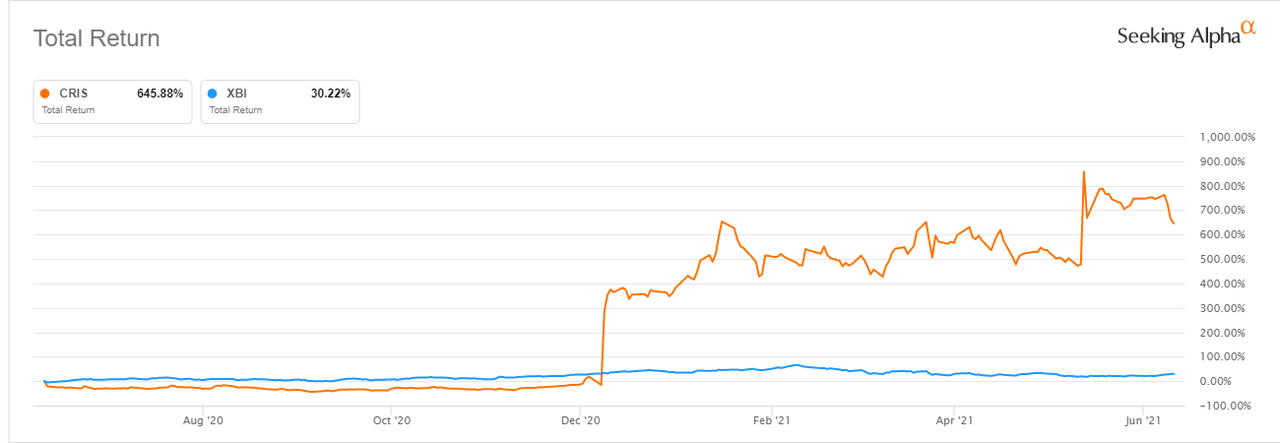
- After a more than a sevenfold rise in value over the past year, Curis (NASDAQ:CRIS) appears to be on track to record its biggest one-day loss in more than a decade.
- Before the market opened today, the company released updated data from its ongoing Phase 1/2 study for CA-4948 in patients with acute myeloid leukemia (AML) or high-risk myelodysplastic syndromes (MDS).
- The trial looking to select the recommended Phase 2 dose involved 22 patients (11 with high-risk MDS, 11 with AML) across dose cohorts of 200 mg BID, 300 mg BID, 400 mg BID, and 500 mg BID.
- Even though with no dose-limiting toxicities (DLT), the company has selected 300mg BID as the recommended dose, safety concerns have appeared in higher dose levels.
- In the 400mg and 500mg cohorts 13% and 66% of patients experienced DLT, respectively, the Curis announced referring to its presentation at European Hematology Association 2021 Virtual Congress (EHA).
- In December, the company had only begun enrollment in the 400 mg BID cohort as no DLTs were observed in seven DLT-evaluable patients at lower dose levels.