Gilnature/iStock via Getty Images
- Phathom Pharmaceuticals (NASDAQ:PHAT) has submitted additional stability data to the US FDA in support of its New Drug Application for the erosive esophagitis asset vonoprazan.
- The six-month stability data was needed to satisfy a requirement in a Complete Response Letter issued in February by the agency.
- The new data indicates that the level of a nitrosamine drug substance related impurity, N-nitroso-vonoprazan, was well below the acceptable limit.
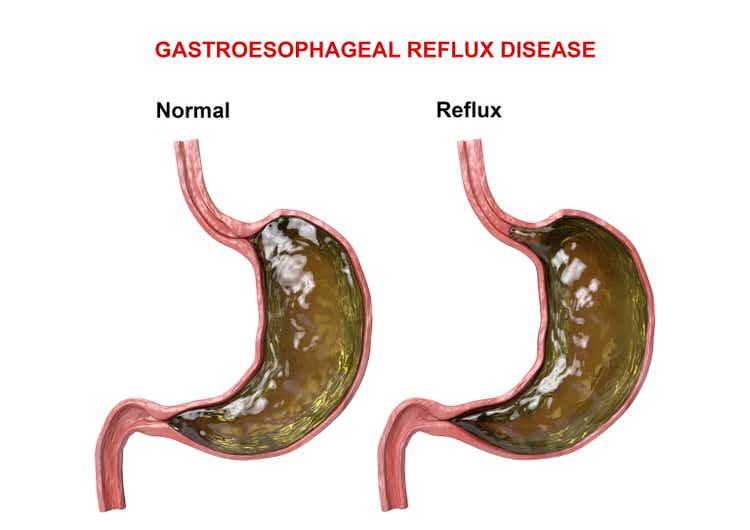
- The FDA accepted Phathom's (PHAT) resubmitted NDA in June. The potassium-competitive acid blocker for erosive gastroesophageal reflux disease has an FDA action date of Nov. 17.