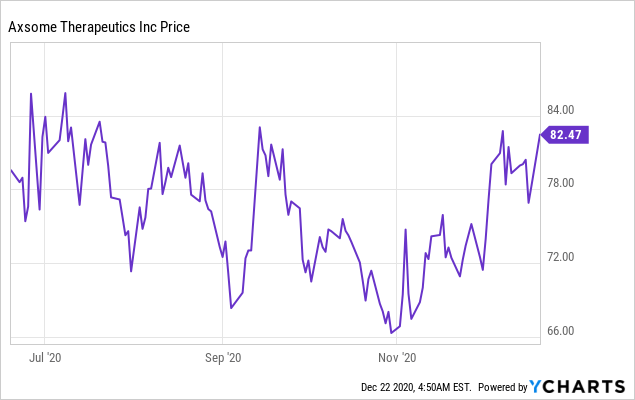
Axsome Therapeutics (NASDAQ:AXSM) has traded between about $65 and $85 the past six months, with recent positive data from AXS-05 providing only some boost to the stock. I take a look at what is ahead for AXS-05 in 2021 that might bring a boost to AXSM.
 Data by YCharts
Data by YCharts
Figure 1: Past six months of AXSM trading.
AXS-05 developments
AXSM announced positive data from a series of trials of AXS-05 (bupropion/dextromethorphan) in a range of depression related indications in December which has provided a bit of a rally from the low $70s to the low $80s, but three positive results in depression would normally get a company further than that, right?
Figure 2: Screen capture of AXSM news releases in December. Source: AXSM website.
Those following the company more closely will know AXSM already produced positive data from double-blind studies containing a placebo control (GEMINI) and active comparator (ASCEND) in major depressive disorder (MDD). Further, AXSM already produced results from a double-blind study of AXS-05 vs. bupropion in treatment resistant depression (TRD) that failed to hit the primary endpoint (p=0.12), despite impressive results on secondary endpoints.
Figure 3: Three of AXSM's previous trials of AXS-05. The major action for AXS-05 in depression has passed, results from ASCEND and GEMINI in 2019, and STRIDE-1 in 2020. Source: September corporate presentation.
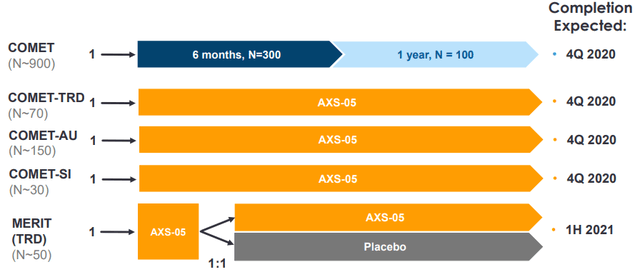
When we look at the more recent trials with AXS-05, the COMET-TRD, COMET-AU (antidepressant unresponsive) and COMET-SI (MDD with suicidal ideation) have produced results but were single arm studies, as is the long-term trial COMET itself. AXSM announced safety results from the long-term COMET trial along with results from COMET-AU on December 1. It is the MERIT study in TRD, a relapse prevention study, that has the chance of producing something compelling.
Figure 4: Overview of more recent AXS-05 studies. Given their single-arm design, it isn't surprising that the COMET-TRD/AU/SI studies have only produced some movement in the share price. Source: September corporate presentation.
Were MERIT to succeed, AXSM could trade up quite a bit. In concert with the success AXSM has had in other studies and the data on secondary endpoints in STRIDE-1, the company might consider running another study in TRD like STRIDE-1. I think if another study like STRIDE-1 were run, and succeeded, then in concert with positive data from MERIT, approval in TRD would be highly likely. That is a lot of "ifs," but the approval of Merck's (MRK) Spravato provides a potential example (see quote below) of how one successful short-term study, and one relapse-prevention study could be considered suitable for approval in TRD. That is opposed to the usual two short-term studies that are normally required in depression for demonstration of efficacy plus a long-term study, such as a relapse prevention study, to demonstrate durability.
"In one of the short-term studies, Spravato nasal spray demonstrated statistically significant effect compared to placebo on the severity of depression, and some effect was seen within two days. The two other short-term trials did not meet the pre-specified statistical tests for demonstrating effectiveness. In the longer-term maintenance-of-effect trial, patients in stable remission or with stable response who continued treatment with Spravato plus an oral antidepressant experienced a statistically significantly longer time to relapse of depressive symptoms than patients on placebo nasal spray plus an oral antidepressant."
- FDA comments on Spravato approval, March 5, 2019. Emphasis mine.
An alternative possibility is that with positive results from MERIT, AXSM might go no further in TRD, simply considering TRD a subset of MDD and allowing physicians to view data from MERIT as an example of how AXS-05 performs in that subset. Certainly comments on the recent earnings call suggest that is a possibility.
"... with regard to TRD, remember that the label that we'll be filing for is MDD, which is the broadest indication and that includes all subsets of major depressive disorder, which includes patients who failed one antidepressants, or patients who failed two or more antidepressants, who would - who may be classified as a treatment resistant, as well as in patients who have suicidal ideation, et cetera.
So we have a broad label, which captures the entire spectrum of MDD patients. And the point of the MERIT trial, as well as the COMET sub studies is to provide data to clinicians in terms of how the product performs in these various populations."
- Comments from AXSM CEO, Herriot Tabuteau, Q3 '20 earnings call.
A run-through of the rest of the catalysts
AXSM is more than AXS-05 in depression related indications, so with a view to going long sometime soon or in early 2021, I had a look at what was on tap.
Figure 5: AXSM catalysts as of November 2020, note COMET-AU/SI/TRD have already produced results (see Figure 2). Source: AXSM earnings presentation.
Starting with NDA submission of AXS-05 for MDD in January 2021, I don't see this as doing much for the stock; although names can run up closer to PDUFA dates, AXSM might run a little when the NDA is (hopefully) accepted for substantive review two months after that.
In Alzheimer's disease (AD) agitation, a trial start will do nothing for the stock, in my opinion.
In smoking cessation, an FDA meeting will provide a path forward. AXSM has data from a phase 2 study in 48 patients. The study was a success; AXS-05 was superior to bupropion, but I think the FDA might want one more study, because 48 patients is not a lot. There is the chance the stock runs if FDA comments are particularly positive.
For AXS-07 in migraine, results from MOVEMENT will do little to move the stock; MOVEMENT is an open-label trial and the market barely reacted to results from MOMENTUM which was an active comparator trial producing impressive results. The market doesn't care for AXS-07 much. Perhaps if it is approved and starts selling well, the market might change its mind.
For AXS-12, a trial start will do nothing, in my opinion, that isn't much of a catalyst.
For AXS-14, if the FDA says AXSM could file for approval in fibromyalgia or needs only supportive studies, then the stock could run. A summary of FDA comments are often released to a company after a meeting (this can take a month) and AXSM would likely issue a press release at that point, noting how well the meeting went.
Summing up
Recent catalysts, such as the readout from several of the COMET studies, have been a little soft, so the reaction in the stock has been a little muted. That could change with MERIT, which likely represents a more impactful catalyst. Press releases concerning the outcome of FDA meetings concerning AXS-05 in smoking cessation and certainly AXS-14 in fibromyalgia also represent potential catalysts for AXSM in H1 '21. Given these three meaningful catalysts are on tap for AXSM in H1 '21, I view the name as a potential long. AXSM had cash of $204.4M at September 30, 2020 and a loan facility of $225M which together it expects to provide funds into 2024. As such, concerns over funding are not likely to come into play in 2021.
The risks of any long in AXSM are several fold, a few of which are worth mentioning here. Firstly, MERIT has an estimated enrolment of 50 patients, which could make achieving statistical significance challenging. Relapse-prevention studies can sometimes be stopped early if they work so well (not likely here, enrolment might be fairly rapid), but 50 patients isn't a lot. On the other hand, AXS-05 only has to beat placebo, so that mitigates risk quite a bit. Secondly, FDA comments on AXS-05 for smoking cessation and AXS-14 for fibromyalgia might be less than encouraging. If a lot of additional work is required, AXSM could trade down. Lastly, AXSM might announce delays in the timeline of any of its trials or regulatory submissions, causing the stock to fall.