Risk come from not knowing what you're doing - Warren Buffett
Author's Note: This is an abbreviated version of an article originally published in advance on April 22, inside Integrated BioSci Investing for our members.
In biotech investing, the old adage of high-risk high rewards rings true. That is to say, it is much riskier for you to invest in clinical-stage companies. After all, failed clinical data of such companies can cause their stocks to crash. Nonetheless, positive data reporting and pipeline advancement over the years can boost the share price several folds higher. If you have an edge in forecasting clinical outcomes, there is nothing more lucrative than early-stage biotech investing.
By having an edge in forecasting, you can mitigate the extreme risk in clinical-stage investing and thereby find your unicorn biotech. On that note, I want to bring to your attention a stock dubbed Avidity Biosciences (NASDAQ:RNA). In this research, I'll feature a fundamental analysis of Avidity and share with you my expectation of its upcoming binary catalyst that can "make or break" this innovator.
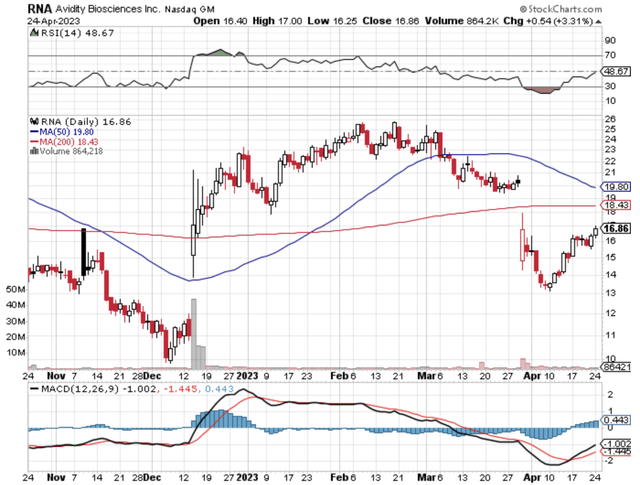
Figure 1: Avidity chart
About The Company
As usual, I'll present a brief corporate overview for new investors. If you are familiar with the firm, I recommend that you skip to the next section. Operating out of San Diego, California, Avidity is focused on the innovation and commercialization of RNA therapeutics to serve unmet needs in rare conditions.
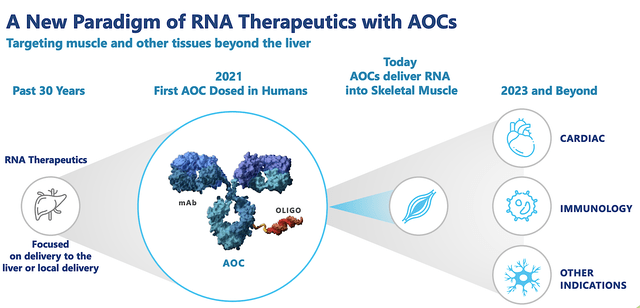
Powering by its Antibody Oligonucleotide Conjugate (i.e., AOC) platform, Avidity is advancing three intriguing RNA medicines (AOC1001, AOC1044, and AOC1020) for Myotonic Dystrophy Type 1 (i.e., DM1), Duchenne Muscular Dystrophy (DMD) and Facioscapulohumeral Muscular Dystrophy (FSHD), respectively.
Figure 2: Therapeutic pipeline
Antibody Oligonucleotide Conjugates Technology Platform
Shifting gears, let us analyze Avidity's AOC technology platform because it underlies upcoming success. Essentially, your AOC drug links an antibody with an oligonucleotide (i.e., either a SiRNA or PMO). Like a smart missile, that antibody carries the whole molecule to the specific target with high precision.
The oligonucleotide then binds to the DNA and thereby exerts its "silencing" effects to prevent the transcription of certain "problematic" DNA sequences into RNA. In other words, this technology platform advances drugs that work on the "central dogma" of biology.
Figure 3: Science/medicine of an AOC drug
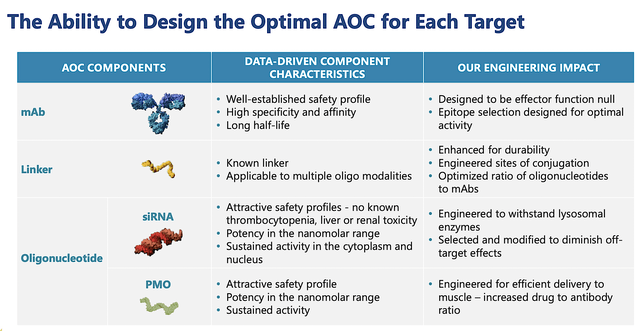
Now, this is an ingenious approach due to the various advantages of an antibody and the siRNA/PMO as shown below. That is to say, there is a tremendous amount of safety/efficacy data on antibodies and PMO/siRNA. Putting them together in a combo, you can imagine it'd unlock their therapeutic synergy. At least, that's the idea behind AOC drugs.
Figure 4: AOC's advantages
Myotonic Dystrophy Type 1(DM1)
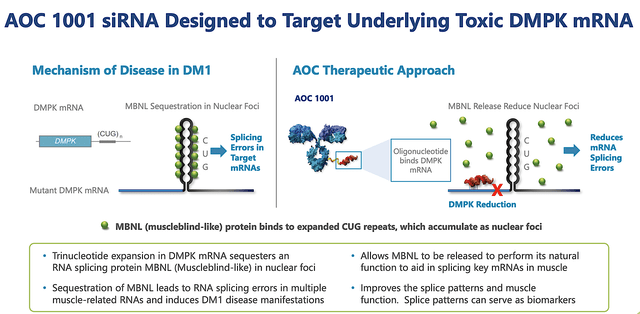
Assuming this is the most fruitful franchise, you should go over the underlying science/medicine of DM1 for you to better appreciate AOC1001. As a progressively lethal genetic condition afflicting over 40K patients in the US, DM1 is caused by triplet repeats in the DMPK gene which interferes with the gene's function.
As a ramification, the disrupted DMPK gene becomes malfunctioned and thereby causes devastating effects on the muscles throughout the body. Consequently, there is increased muscle wasting and weakness. Patients suffering from this condition often have prolonged muscle contractions (i.e. myotonia). Therefore, they are not able to relax their muscles after use. For example, patients might not be able to release their grip on the door handle.
Figure 5: DM1 statistics
AOC1001 for DM1
You can see there is currently no approved therapy for DM1. As such, you can bet that the demand for any treatment is extremely robust. In filling the heightened demand, Avidity is advancing AOC1001 for DM1. As you can appreciate, AOC1001 works by silencing the triplet repeats in the DMPK gene. Therefore, it allows the cellular machinery to splice (i.e., cut) out the repeats in the DMPK's gene mRNA for it to function normally.
Figure 6: Mechanism of action of AOC1001
Early Data Supporting Efficacy
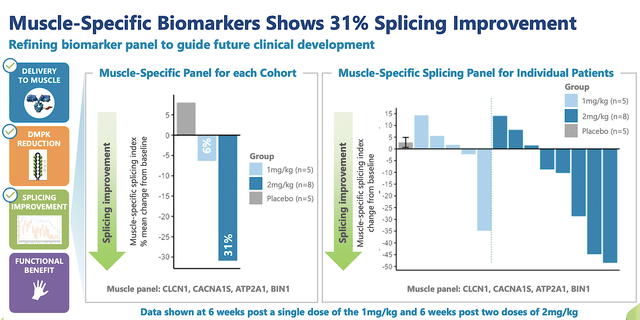
As you know, all the sound underlying science/medicine is meaningless unless the drug can generate robust clinical efficacy. In an early clinical study, AOC1001 demonstrated a 31% splicing improvement based on muscle-specific biomarkers as shown below.
Figure 7: Strong early data results of AOC1001
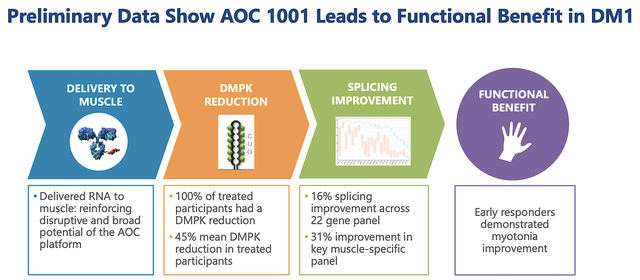
More important than the biomarker data, you can see that AOC1001 treatment leads to functional benefits for the patients. That is to say, early responders demonstrated myotonia improvement. Precisely speaking, they moved their hands better.
Figure 8: Functional improvement for patients taking AOC1001
Partial Clinical Hold
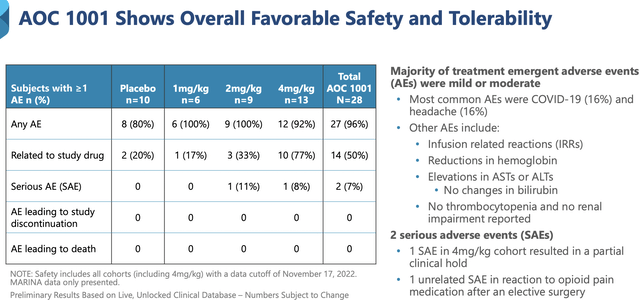
Overall, AOC1001 was safe and well tolerated. Nonetheless, there was one patient death in September last year which was due to a brain bleed. As you can see, this occurred in the patient who took the highest dose.
Interestingly, the investigator could not find any direct causation because AOC1001 does not cross the blood-brain barrier. In an effort to ascertain safety, the FDA currently placed a "partial hold" on the Phase 1/2 (MARINA) trial. As a ramification, patients already on the trial can continue to receive the drug. However, no new patient can be enrolled. Commenting on this obstacle, the President and CEO (Sarah Boyce) remarked,
The safety of patients is our top priority. We are grateful to the participants in our clinical trials and their families, as well as the broader DM1 community, for their ongoing trust and collaboration. We share the urgency for a treatment for people living with this devastating disease with no approved therapies. Our team is focused on resolving the partial clinical hold and advancing AOC 1001 to a pivotal trial as quickly as possible. We are continuing to work diligently with the FDA and remain very confident in AOC 1001 and its benefit/risk profile. We will continue to share emerging data with the FDA as we conclude the MARINA trial. We look forward to reporting safety and functional top-line data from the MARINA trial at AAN next month.
Figure 9: AOC's safety profile
Typically, you can expect a partial clinical hold to be resolved around 6 months. As you can see, AOC1001 already surpassed that point. Nevertheless, if the upcoming MARINA topline safety/efficacy data report is positive, it can prompt the FDA to remove the partial hold in the ensuing months.
Financial Assessment
Just as you would get an annual physical for your well-being, it's important to check the financial health of your stock. For instance, your health is affected by "blood flow" as your stock's viability is dependent on the "cash flow." With that in mind, let us assess the 4Q2022 earnings report for the period that ended on December 31.
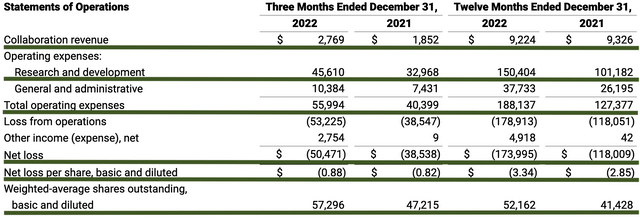
As follows, Avidity procured $2.7M in revenue compared to $1.8M for the same period a year prior. Notably, the revenue comes from its collaborative partnership with Lilly. Given that Avidity is still in its early clinical stage of investigations, you should focus on other more meaningful metrics.
That being said, the research and development for the respective periods registered at $45.6M and $32.9M. I viewed the 38.6% R&D increase positively because the money invested today can turn into blockbuster profits tomorrow. After all, you have to plant a tree to enjoy its fruits.
Additionally, there were $50.4M ($0.88 per share) net losses compared to $38.5M ($0.82 per share) net declines for the same comparison. As you can see, the increased R&D cut into the bottom-line earnings. As the company further advances its pipeline, you can anticipate the bottom line depreciation to widen.
Figure 10: Key financial metrics
About the balance sheet, there were $610.7M in cash, equivalents, and investments. Against the $55.9M quarterly OpEx, there should be adequate capital to fund operations into 3Q2025 prior to the need for additional financing. Simply put, the cash position is robust relative to the burn rate.
Potential Risks
Since investment research is an imperfect science, there are always risks associated with your stock regardless of its fundamental strengths. More importantly, the risks are "growth-cycle dependent." At this point in its life cycle, the most immediate concern for Avidity is whether AOC1001 would post positive MARINA data soon. Moreover, there is a chance that AOC1001 would not regain its partial hold removal. That aside, the other franchises (AOC1021 and AOC1044) might deliver subpar clinical results.
Concluding Remarks
In all, I issued a highly speculative buy recommendation on Avidity with a 4.5/5 stars rating. Avidity Biosciences is an interesting company that is powered by the intriguing technology platform, AOC. You can expect that this novel form of RNA medicine will continue to gain market traction.
With differentiation, AOC is poised to garner significant market success as it serves to fill the unmet needs in orphan conditions. The upcoming clinical binary event (i.e., the MARINA trial report) will decide Avidity's fate soon. If positive, you can expect the shares to rally. Else, this stock is likely to tumble significantly from its current market valuation.
I hope you enjoyed reading this general free article. Members of our Private Investment Group, Integrated Biosci Investing get EXCLUSIVE catalyst-focused research on this company in advance. Our catalyst-forecasting EDGE allows members to profit BIG amid this bear market. As the #1 Biotech Group for Growth Investing and Power Catalysts Trading, you can leverage our expert EDGE to know biostocks would fly soon.
Now, you can emulate Warren Buffett by making two solid decisions a year. Make your first decision by hitting that "FOLLOW" button on top for the latest updates. Make your second great decision by getting your FREE 2-week trial NOW!