- Nevro Corp. (NVRO +5.4%) continues to trade higher after the company announced the FDA approval of its Senza Spinal Cord Stimulation (SCS) system as a treatment of chronic pain linked to Painful Diabetic Neuropathy (PDN).
- Commenting on the results, Piper Sandler analyst Adam Maeder notes that the FDA greenlight offers a “meaningful multi-year growth driver to the NVRO story.”
- “While SCS volumes are admittedly taking a bit longer than expected to snapback, the key tenets of our NVRO thesis remain intact,” Maeder argues.
- With the company planning to initiate the commercial launch activities of Senza System in the U.S. immediately, the approval signifies an “important milestone,” the analyst says, citing a multi-year first-mover advantage for the company in the sizable end market.
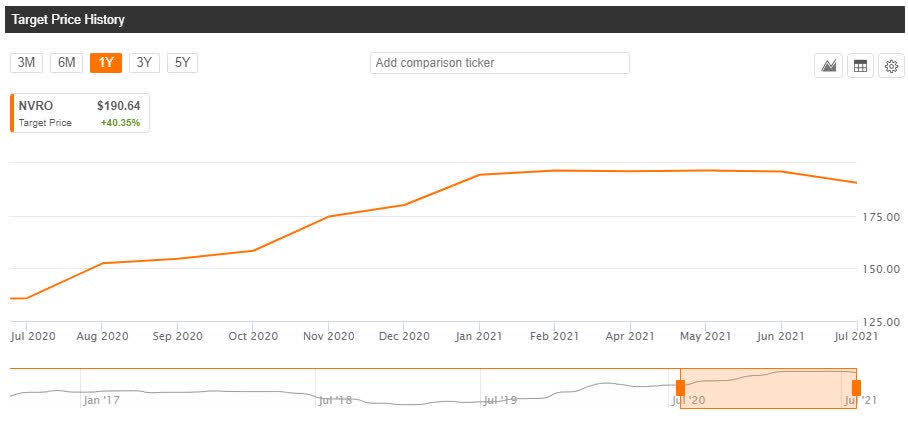
- Piper Sandler has an overweight rating on Nevro, and the $200 per share price target implies a premium of ~38.7% to the last close.
- See how the average price target in Wall Street has changed for Nevro over the past 12-month period.