tumsasedgars/iStock via Getty Images
Thesis
Annovis (NYSE:ANVS) should report data from its Phase 3 trial in Parkinson’s [PD] and Phase 2/3 trial Alzheimer’s disease [AD] by January and March 2024 respectively.
The ongoing studies are symptomatic. However, Annovis claims that Buntanetap is disease-modifying as it inhibits the production of neurotoxic proteins. These proteins are involved in normal cellular processes, and recent scientific research considers only aggregated forms of these proteins neurotoxic.
I find the remarkable short-term efficacy both in AD and PD hard to align with the drug’s proposed disease-modifying mechanism of action. The stakes are high in my eyes. Improving symptomatic standard of care has its own standards both in AD and PD, compared to a potentially disease-modifying drug. The knowledge whether a drug show symptomatic effects or truly modifies the course of the disease is therefore of importance, even more so as an allegedly disease-modifying drug will currently report on symptomatic trials.
Annovis may raise money around at least one of these events. In the past, certain events around which capital has been raised have led to investor criticism. Initially positive stock reactions have often been tempered subsequently.
Company
Introduction
Annovis is a biotech company focused on the treatment of neurodegenerative diseases. Its lead compound Buntanetap is in trials for the treatment of Alzheimer’s [AD] and Parkinson’s disease [PD].
At the time of writing, Annovis has a $80 million market cap. This is the five-year trajectory of Annovis’ stock.
Five-year stock chart Annovis (Google)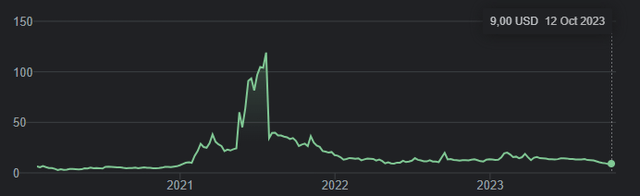
In 2021, after having gone up to an almost $1 billion market cap, the stock has fallen drastically shortly thereafter on the back of data and severe criticism covered extensively by Seeking Alpha analyst C.C. Abbott in four separate articles, after two initial bullish analyses she had also authored. Together they bring a stringent analysis of Annovis’s reporting in 2021, which I would suggest readers make part of their due diligence.
Annovis’ most pivotal times may be upon us. With market sizes of $4 billion for Alzheimer’s and $4.5 billion for Parkinson’s respectively which are both expected to grow massively, and historical gains and volatility of other AD companies in the framework of positive results, there is an interest for investors to make their analysis as diligently as possible.
Buntanetap
Standard of care in AD and PD
The standard of care in AD consists of acetylcholinesterase inhibitors, i.e. drugs acting upon the neurotransmitter acetylcholine. These are symptomatic treatments, the first one of which has been approved in 1993, and the most known is Donepezil, approved in 1996. Donepezil produces about a 2-point improvement [i.e. decline] on the Adas-Cog rating scale over six months.
The years 2021 and 2022 saw the approval of the first truly disease-modifying drugs in AD, anti-amyloid antibodies Aduhelm and Leqembi. They reduce cognitive decline by about 30%.
In PD, the symptomatic treatment carbidopa/levodopa is most prescribed, improving patient’s ON time. There are no disease-modifying drugs for PD yet.
Buntanetap’s enantiomer posiphen
Buntanetap or posiphen is the pure enantiomer of phenserine. Phenserine had been in development for AD and had been touted as the next-generation acetylcholinesterase inhibitor with a dual MoA including non-cholinergic properties, but its drug development has been abandoned +15 years ago. These were the cognitive study results at 26 weeks on Adas-Cog, showing a treatment divergence of -1.9 points with placebo at 26 weeks for the 15mg dose. Such result would be in line with Donepezil’s results [see below].
Phenserine study results 26 weeks (J Alzheimer's Dis. )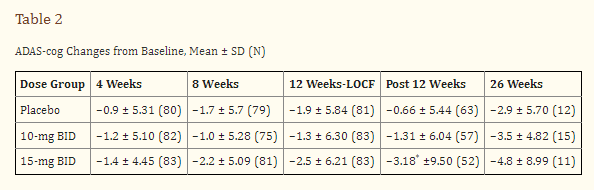
As a pure enantiomer of phenserine, posiphen is its molecular mirror image. One of the publications referred to by Annovis considers that posiphen does not have a cholinergic MoA, but another one does consider that posiphen’s MoA may be cholinergically mediated.
Buntanetap’s mechanism of action
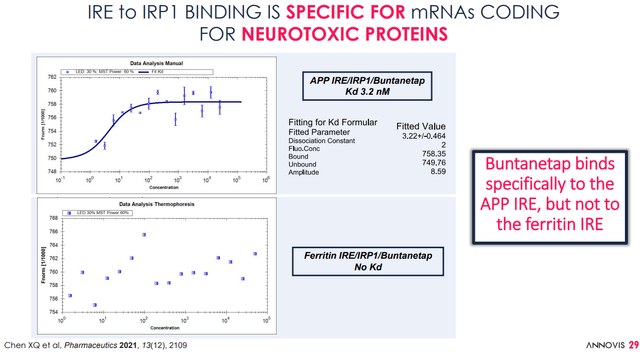
For Annovis, the MoA of Buntanetap would lie in inhibiting the production of neurotoxic proteins, such as amyloid-beta, tau, α-synuclein and TDP-43, by Buntanetap binding to the IRE of the amyloid precursor protein APP, which would be specific for coding of these proteins.
Buntanetap proposed MoA (Annovis corporate presentation) Buntanetap proposed MoA slide 2 (Annovis corporate presentation)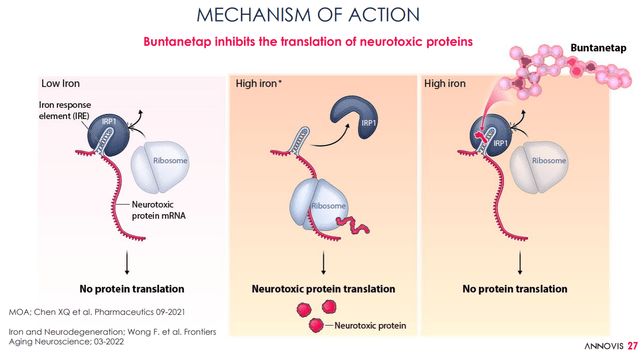
Buntanetap’s synergistic action on all such proteins is claimed to lead to its efficacy, as the removal of one neurotoxic protein would only result in minimal effect.
Buntanetap inhibits production of proteins (Annovis corporate presentation)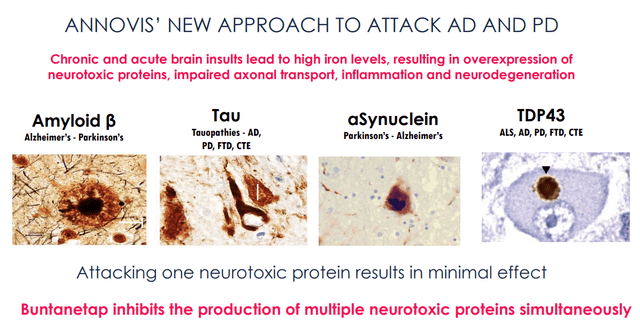
All of these proteins are indeed involved in major neurodegenerative diseases.
I have two remarks here.
- Buntanetap and phenserine both act on IRE sequences in the 5' untranslated region of APP mRNA to inhibit APP protein synthesis, reduced APP and Aβ in animal models, and block translation of α-synuclein mRNA.
- Though Annovis claims one should not argue about whether these proteins are toxic [R&D Day, min. 6], these proteins actually have functions. Amyloid is involved in protection from infections, repair of the blood-brain barrier, recovery from injury and synaptic function. Alpha-synuclein is involved in synaptic function. Tau is a structural element. TDP-43 is involved in DNA repair. APP has a trophic function in the nervous system. Current scientific insights do not consider these proteins toxic as such but in their aggregated forms which form over time. This holds true for amyloid and tau, primarily involved in AD. Below is a slide from Acumen Pharmaceuticals’ (ABOS) scientific page with regard to amyloid aggregates.
Different amyloid aggregates slide (Acumen Pharmaceuticals corporate presentation)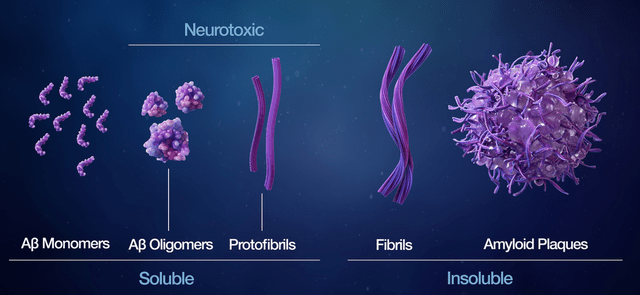
It also holds true for α-synuclein, primarily involved in PD, huntingtin, and TDP-43.
Alpha-synuclein cascade hypothesis (Front. Neuroscience)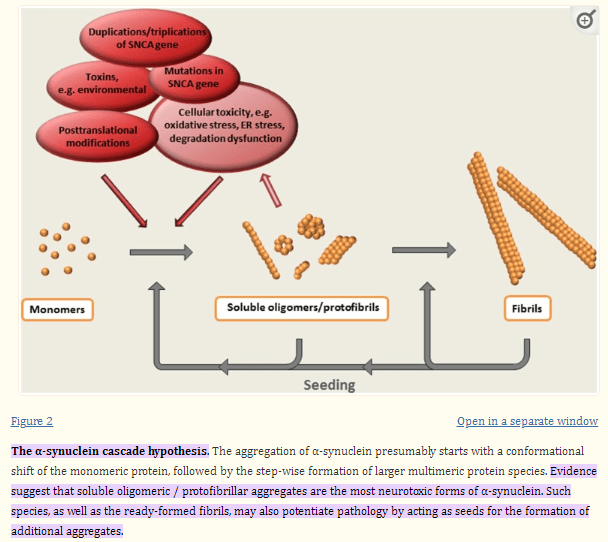
ADCS and AD/PD trials
Annovis, previously called QR Pharma, has done several short-term trials with posiphen over the course of the past years.
As QR Pharma, Annovis’ ADCS study had started in 2016, to test 24 patients with early AD to receive 60, 120, or 180 mg daily or placebo for 23 to 25 days. Annovis reported cognitive data from that trial only in 2023.
The AD/PD trial, results of which had led to the meteoric rise and fall of Annovis’ stock in 2021, had started in 2020. This was a two-part study. The first part was composed of 28 patients divided into two subgroups: 14 with early PD and 14 with early AD. The second part was composed of 40 patients with early PD. Dosing was different; the first group received 80 mg per day, the second group was randomly assigned to receive 5 mg, 10 mg, 20 mg, or 40 mg per day.
Biomarker data
The biomarkers of a truly disease-modifying drug should give a good impression on expected efficacy.
Annovis reported the following data on biomarkers in patients taking the high dose of 80 mg per day. This may not be the desired dose for either PD or AD, as Annovis claims dosing of patients at these levels is too high and dosing of 10/20 mg would be more appropriate. Annovis’ CEO stated during Annovis’ R&D Day:
In our case, the higher the dose, the worse. [...] At high dose, the animals are slightly agitated.
Biomarker changes 28 days slide 1 (Annovis corporate presentation )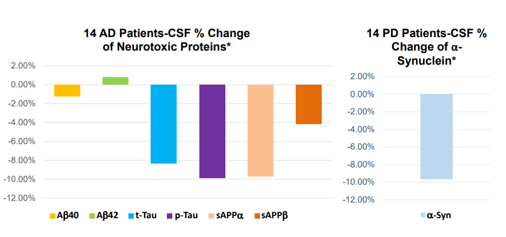
Biomarker changes 28 days slide 2 (Annovis corporate presentation )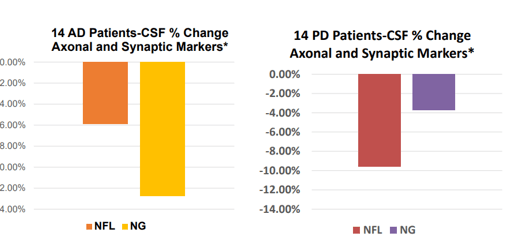
Biomarker changes 28 days slide 3 (Annovis corporate presentation)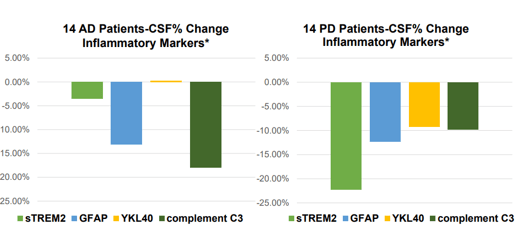
These results at 80 mg per day are moderate if one compares them to, for instance, those of Cassava Sciences (SAVA), over the same 28-day study period. Cassava Sciences has its own share of scrutiny, which the CEO of Annovis agrees to. Nonetheless, the data is out there. For Annovis, numbers are estimates based on the above charts.
Comparison results Annovis / Cassava Sciences 28 days (Own work)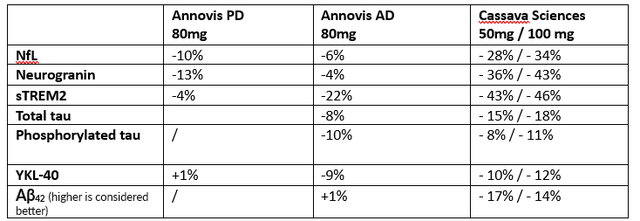
On average, Cassava’s results appear about 3-4 times as strong as those of Annovis, and what’s more, Cassava’s biomarkers massively improved at the six-month timepoint. My view here is Buntanetap has a moderate effect on biomarkers at 28 days, even at high dose.
For NfL, a biomarker of neurodegeneration which has been retained for accelerated approval in ALS after seeing a 55% decrease over 28 weeks, Buntanetap’s data at 80mg per day may not be impressive. For reference, INmune Bio’s (INMB) Xpro had reported an 84% reduction of this biomarker over the course of three months in AD patients, and Cassava Sciences’s simufilam a 55% decrease over six months. Given their respective MoA’s, I assume the latter like BioVie’s (BIVI) NE3107 are drugs with true disease-modifying potential. During the AdCom meeting for Brainstorm Cell Therapeutics’ (BCLI) Nurown, the FDA questioned whether a 8.9% reduction over placebo at 20 weeks would be sufficient to indicate a treatment effect. Annovis’ NfL data scores in that range.
In summary, Annovis’s biomarkers fail to impress me and I find the efficacy reported by Annovis at 28 days hard to reconcile with biomarkers reported by other companies I have covered in the neurodegenerative space.
Buntanetap for Parkinson’s disease
Results so far
Annovis’ Phase 2 trial has data from 54 patients: 40 at 80 mg, and then 14 patients spread out over 5 groups: placebo, 5mg, 10mg, 20mg or 40mg, treated during 28 days. Annovis reported data on the MDS-UPDRS scale and on the WAIS coding test.
MDS-UPDRS scores 28 days (Annovis presentation AD/PD conference)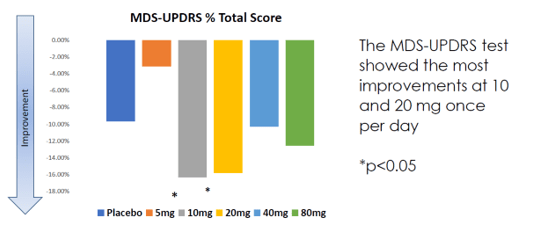
The MDS-UPDRS scale, widely used for assessing the severity and progression of PD, also includes non-motor aspects of daily living [part I], tremor and rigidity [part III], particularly tremor, in PD. It is not excluded that cholinergic drugs for PD with a differentiated or double MoA may result in better overall scoring compared to placebo.
The MDS-UPDRS results show better efficacy at 10mg and 20mg. The suggestion that lower dosing would be more effective was confirmed by Annovis’ CEO [min. 34], although I find the remark that higher doses make the animals agitated hard to align with the proposed MoA, as that could relate to potentially receptor-overtriggering.
If we do dose that reduces tau, amyloid, α-synuclein at 50%, then the animals are slightly agitated.
At 3-10% decrease, and that’s true for α-synuclein, that’s true for tau, that’s true for A-Beta, that’s true for APP, I see beautiful efficacy in animals. And guess what, we see the exact same thing in humans. The lower the dose, the better the efficacy, the less we see a decrease. You saw a 50% decrease at 180 meg, at 180 meg, patients show no efficacy, none, zilch, zero. They show it at 60 and 80, but remember, we saw efficacy at 80 in Parkinson’s, but then we saw better efficacy at 10 or 20. […] We see a very strong reverse U-shape.
WAIS coding results 28 days (Annovis presentation AD/PD conference)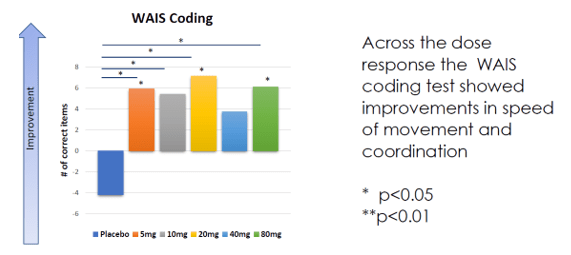
The coding subtest of the WAIS IV test, a test used to assess cognitive strength, tests coding speed. It is, I believe, a measure that has little to do with PD’s main issue, namely motor function. I quote the following statement from Annovis’ CEO, when asked about the probability of success in PD [min. 23]:
We believe that the most likely to function would be dementia in Parkinson’s. Whether we have any effect on OFF time, we really don’t know.”
ON time is the period of time when a patient feels the effect of carbidopa/levodopa, the symptomatic standard of care in PD. Carbidopa/levodopa has no effect on OFF time. OFF time refers to the time period when no levodopa/carbidopa is taken. A truly disease-modifying treatment allegedly removing neurotoxicity should show its potential at that point.
As to the remark that Buntanetap would most likely function in dementia in PD, Parkinson’s Disease Dementia only occurs on average 10 years after Parkinson’s diagnosis and hence at a very late stage.
Phase 3 trial data in January 2023
The ongoing symptomatic Phase 3 study has enrolled 523 patients, spread into three groups: placebo, 10 mg/day and 20 mg/day, for 6 months.
Development pipeline PD (Annovis corporate presentation)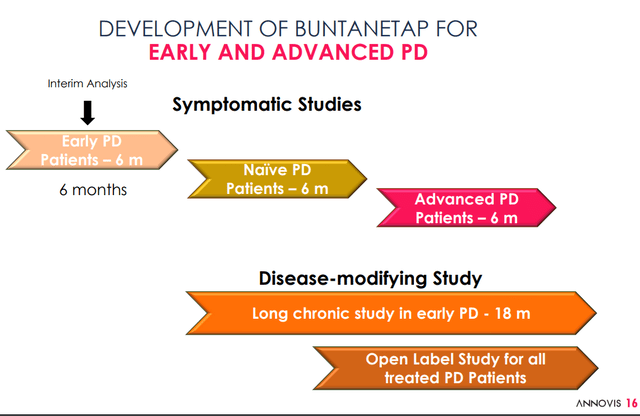
Phase 3 trial design (Annovis corporate presentation)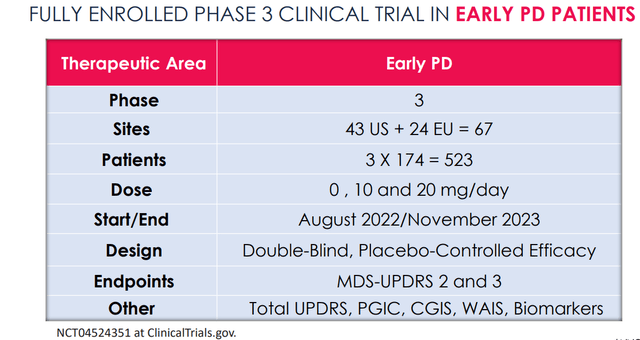
There has been an interim analysis which has led to much confusion for investors, as they have been led to believe that data from this trial would be presented at a major conference. Seeking Alpha analyst Anders Research provided good coverage of that moment. Annovis’ CEO took the blame for that during Annovis’ R&D Day and on this website, but the damage had been done in my eyes. Annovis’ CEO stated the following during the same R&D Day:
Now, realistically all we really know today is that our drug works at one month, that was the Phase 2, and two months, that is the interim analysis, and now we need to see whether it works after six months.”
However, no data were communicated at two months, hence it is not known whether the drug works at two months, as stated earlier [min. 4]:
Unfortunately it was blinded, we did not want to ruin the study, and really all it shows us is go/no-go.
The symptomatic Phase 3 study in PD will report data in January 2023. To find out more about Buntanetap’s MoA and efficacy, I am particularly curious about biomarkers and the trajectory of the data. Annovis may however not share these together with topline data.
Annovis has not made any suggestions about accelerated approval and on the basis of the biomarker data to date, I do not think that is a real possibility. That means any possible approval would need to await the results of the disease-modifying study, and is probably for some time in 2026 at the soonest. Meanwhile, Annovis will need a lot of cash to conduct such a trial.
Buntanetap for Alzheimer’s disease
Results so far
In AD, the following findings were reported.
AD cognitive efficacy reported so far (Annovis corporate presentation)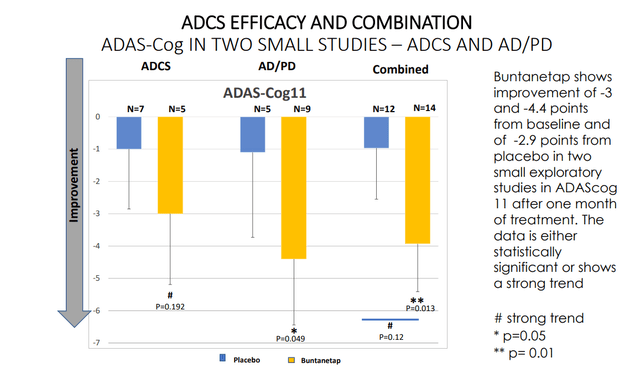
There is a systematic placebo effect which one would have to discount, but otherwise, the onset and impact of these findings is remarkable.
As for the ADCS results, though Annovis’ chart shows measurements of Adas-Cog11, the study results on clinicaltrials.gov mention measurements of Adas-Cog12 which appear to differentiate from the above, with a higher placebo response. On Adas-Cog12, treated patients apparently scored -0.55 and patients on placebo scored -1.57. In that trial, the MMSE rating scale was also used, reporting that placebo improved by 1.14 points, the 60mg group declined by 1.4 points, the 120mg group improved by 1.2 points, and the 180mg group declined by a point again. Less efficacy is seen in the ADCS trial, which had 60, 120 and 180 mg per day, i.e. 120mg on average, whereas the AD/PD study had 80 mg/day as dose. As mentioned before, Annovis’ CEO had stated there was no efficacy seen at 180mg, though I believe unpooled data for the ADCS study were not communicated.
Most stunningly, however, is the -4 point improvement on Adas-Cog11 over the course of 28 days as seen in the the AD/PD trial , a result that appears to outperform all other results reported so far. It is for that reason that I remain left with a feeling that Buntanetap’s action may be more symptomatic – directly or indirectly – and hence similar to phenserine than appears.
The supplementary information to the scientific publication covering this trial are shown below. For reference, the MMSE is a 30 point rating scale and the CDR-SB ranges from 0 to 3.
Phenserine cognitive changes 26 weeks (Journal for the Prevention of Alzheimer's Disease)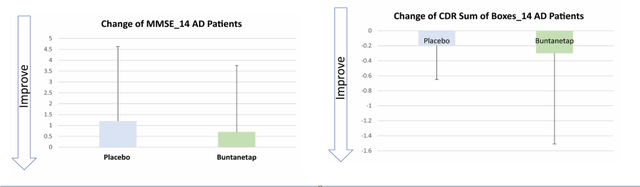
In light of what Annovis reported, Buntanetap may score better than standard of care symptomatic treatment donepezil. Donepezil’s trajectory is interesting, as it peaked at six months only to progress downward from there on.
Donepezil cognitive trajectory (Dement. Geriatr. Cogn. Disord. 1999)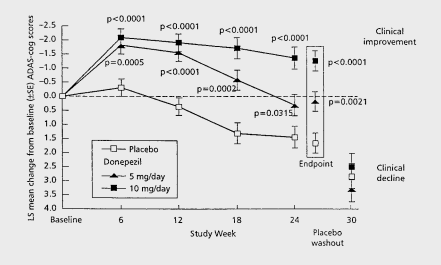
My fear, here, is that these strong results may not last in a disease-modifying study. One could argue that, if Buntanetap would also indirectly act cholinergically, at least it also acts on proteins that aggregated and may become neurotoxic over time. I do not exclude that possibility, but I do not know, nor does Annovis’ CEO apparently, when she considered during Annovis’ [min. 43]:
I’m really low-balling it because I don’t know how the course is going. And of course what we all know is the drug could lose some efficacy as we go along.
I remark, in that regard, that although it is primarily seen as a symptomatic agent, Donepezil has also been reported to have neuroprotective and non-cholinergic mechanisms, as cholinesterase inhibitors have been reported to influence APP metabolism and tau. The same held true for phenserine.
It is still unclear what Buntanetap’s efficacy or trajectory would be, and I hope we see data on this right away when topline data will be presented in March 2024, similar to Biogen’s reporting of Leqembi as shown below, suggestive of disease modification.
Leqembi cognitive trajectory (Biogen PR)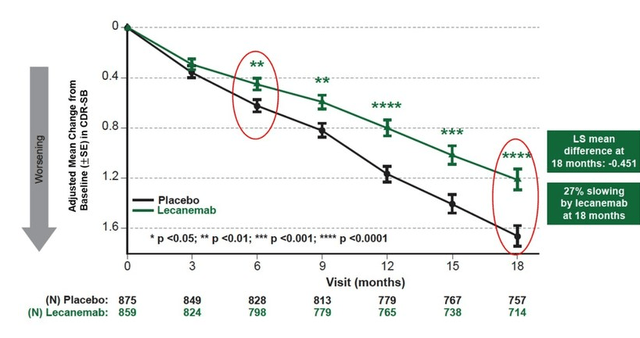
If one would be able to affect cognitive decline, the trajectory over a longer period of time is important. In the case of Donepezil, the decline started again at six months.
I also note that anti-amyloid antibodies are apparently an exclusion criteria for future trials for Annovis [min. 28] though the Alzheimer’s Drug Discovery Foundation considers that combination therapies are key to further success in neurodegenerative diseases.
Phase 2/3 trial results in March 2023
This is Annovis’ progress with regards to the development of Buntanetap for Alzheimer’s.
Buntanetap for AD pipeline (Annovis corporate presentation)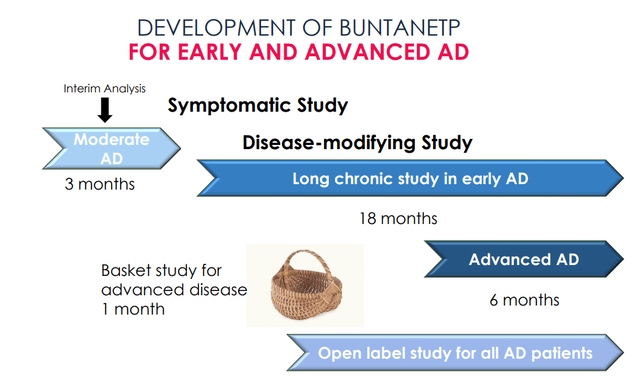
The ongoing symptomatic Phase 2/3 study is another short three-month study which should enroll 320 patients, 230 of which had been enrolled early October 2023, with full enrollment would already be expected by November 2023 which would mean the trial would need to enroll another 90 patients in about two months’ time. Data of that trial should in such case read out in March 2023. As it is a dose response study [min. 24], meaning each patient group will have only 80 patients, this really feels more like a Phase 2 study.
Phase 2/3 trial for AD trial design (Annovis corporate presentation)
Annovis has not given any indications as to how such study could give rise to the possibility for approval. Hence, success in a long 18-month chronic study in early AD would be needed to go for full approval. Realistically, even with Annovis’ fast enrollment, I would not expect results of such study before 2026.
With topline data expected in March 2023, I am again most interested in biomarker results and the trajectory of the patients’ cognitive function.
Financials and metrics
Annovis currently has 9,012,273 shares outstanding, of which 2,755,831 shares are held by insiders, representing 30.58% insider ownership.
Annovis had cash of $15.7 million at the end of the second quarter of 2023, and reported a Q2 net loss of $9.5 million. According to the company, that money would be adequate to finish its two studies, and then have an additional three months. I do not think the current cash level would allow Annovis to have sufficient cash to last it until June 2024, given its last-year’s cash burn of $29 million. Annovis has made it no secret it may raise money around pivotal events such as data readouts. As the next studies are supposed to be 18-month studies testing for disease-modification, I expect Annovis to want to raise a considerable amount of money in the near future.
Risks
Like with any biotech stock, investing is risky. A drug may fail to report statistical significance, the stock may be highly volatile, there is high competitive risk and regulatory authorities need to validate any further progress the company desires. Annovis has two big events coming up, so that caution is in place particularly at this point in time.
Aside from the risks set out above, both PD and AD trials have enrolled patients with PD or AD diagnosis respectively, but did not specifically enroll patients on the basis of their underlying biology. If Buntanetap’s only MoA is truly disease-modifying, this is a risk given the multifactorial character of these diseases. For example, the implication of endotoxins, gut function or mitochondrial dysfunction in PD is widely discussed. A trial enrolling all-comers may put the trial at risk of not reaching statistical significance.
The company is low on cash, and Annovis plans to raise money in the three months following both readouts at the latest [min. 4]. Looking at the planned disease-modifying trials, that dilution may be significant.
Conclusion
Buntanetap’s and Annovis’ progress in both Alzheimer’s and Parkinson’s disease are interesting to watch for me, given the historical price volatility and scrutiny many investors have expressed since the stock’s rise and fall in 2021.
Annovis will soon report data from both a 6-month Phase 3 trial in Parkinson’s and a 3-month Phase 2/3 trial in Alzheimer’s disease. Both studies are symptomatic. With a combined market size of $8.5 billion, expectations of massive growth in both indications, and historical gains and volatility in the case of results perceived as positive, there is an interest for investors to make their analysis as diligently as possible.
Results reported from previous trials are remarkable, though I find the biomarker data hard to align with Buntanetap’s proposed MoA and efficacy reported so far.
The proteins Buntanetap inhibits may not be neurotoxic upon their creation, rather upon their oligomerization. Buntanetap may exert indirect cholinergic activity, and if so, the question is to what degree, given posiphen’s enantiomeric character as compared to the abandoned phenserine, which had improved cognition at 26 weeks by 1.9 points on Adas-Cog at 15mg per day dosing.
The differentiation between symptomatic and disease-modifying is essential. A truly disease-modifying drug in PD should show efficacy during OFF time. A disease-modifying drug in AD should differ from the characteristics of acetylcholinesterase inhibitors, which is why biomarkers matter so much. I am interested in potential effects on OFF time in PD, biomarker results, and the trajectory of possible efficacy in both indications, beyond 6 and 3 months respectively. Much of the latter data will also probably only be reported after long 18-month trial focusing on disease-modification.
Annovis may need to raise a serious amount of money to conduct these trials.
Without excluding Buntanetap’s chance to offer a disease-modifying solution for patients in both indications, I will be standing on the sidelines awaiting further data.
Editor's Note: This article covers one or more microcap stocks. Please be aware of the risks associated with these stocks.